19-Nordehydroepiandrosterone
19-Nordehydroepiandrosterone (19-nor-DHEA), or 19-nor-5-dehydroepiandrosterone (19-nor-5-DHEA), is an estrane (19-norandrostane) steroid which was never marketed.[1][2] It is the combined derivative of the androgen/anabolic steroid nandrolone (19-nortestosterone) and the androgen prohormone dehydroepiandrosterone (DHEA, or more specifically 5-DHEA).[1][2] Related compounds include 19-nor-5-androstenediol, bolandiol (19-nor-4-androstenediol), and bolandione (nor-4-androstenedione), which are all known orally active prohormones of nandrolone.[2][3][4] 19-Nor-DHEA may occur as a metabolite of bolandione and related steroids.[2]
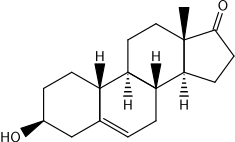 | |
| Clinical data | |
|---|---|
| Other names |
|
| Routes of administration | By mouth |
| Drug class | Androgen; anabolic steroid; progestogen |
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| Chemical and physical data | |
| Formula | C18H25O2 |
| Molar mass | 273.396 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
See also
References
- Poortman J, Vroegindewey-Jie D, Thijssen JH, Schwarz F (July 1977). "Relative binding affinity of androstane and C-19-nor-androstane-steroids for the estradiol-receptor in human myometrial and mammary cancer tissue". Mol. Cell. Endocrinol. 8 (1): 27–34. doi:10.1016/0303-7207(77)90015-6. PMID 881104. S2CID 24190327.
- Uralets VP, Gillette PA (April 2000). "Over-the-counter Δ5 anabolic steroids 5-androsen-3,17-dione; 5-androsten-3β, 17β-diol; dehydroepiandrosterone; and 19-nor-5-androsten-3,17-dione: excretion studies in men". J Anal Toxicol. 24 (3): 188–93. doi:10.1093/jat/24.3.188. PMID 10774538.
- Detlef Thieme; Peter Hemmersbach (18 December 2009). Doping in Sports. Springer Science & Business Media. pp. 137–. ISBN 978-3-540-79088-4.
- Jehuda Yinon (29 December 2003). Advances in Forensic Applications of Mass Spectrometry. CRC Press. pp. 138–. ISBN 978-0-203-99828-1.
Estrogen receptor modulators | |||||||
|---|---|---|---|---|---|---|---|
| ER |
| ||||||
| GPER |
| ||||||
| |||||||
Progesterone receptor modulators | |||||||
|---|---|---|---|---|---|---|---|
| PR |
| ||||||
| mPR (PAQR) |
| ||||||
| |||||||
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.