Metapristone
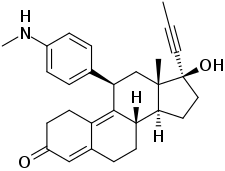
Metapristone (developmental code name RU-42633; also known as desmethylmifepristone) is the major metabolite of mifepristone (RU-486, RU-38486) and a selective progesterone receptor modulator (SPRM) which itself was never marketed.[1][2][3][4] It is formed from mifepristone in the liver by the enzyme CYP3A4 via monodemethylation, and circulates at concentrations higher than those of mifepristone.[1][5] The metabolite retains partial but considerable affinity for the progesterone receptor (PR) and the glucocorticoid receptor (GR) (RBA = 21% and 61% of that of mifepristone for the human forms of these receptors, respectively).[6][1] On the basis of actions that are apparently independent of its hormonal activity, metapristone is being researched as a potential cancer metastatic chemopreventive agent.[2][3][4]
 | |
| Clinical data | |
|---|---|
| Other names | RU-42633; Desmethylmifepristone; 17β-Hydroxy-11β-[4-(methylamino)phenyl]-17α-(prop-1-yn-1-yl)estra-4,9-dien-3-one |
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C28H33NO2 |
| Molar mass | 415.577 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
References
- Heikinheimo O (July 1997). "Clinical pharmacokinetics of mifepristone". Clin Pharmacokinet. 33 (1): 7–17. doi:10.2165/00003088-199733010-00002. PMID 9250420. S2CID 25101911.
- Wang J, Chen J, Wan L, Shao J, Lu Y, Zhu Y, Ou M, Yu S, Chen H, Jia L (March 2014). "Synthesis, spectral characterization, and in vitro cellular activities of metapristone, a potential cancer metastatic chemopreventive agent derived from mifepristone (RU486)". AAPS J. 16 (2): 289–98. doi:10.1208/s12248-013-9559-2. PMC 3933578. PMID 24442753.
- Wang J, Chen J, Zhu Y, Zheng N, Liu J, Xiao Y, Lu Y, Dong H, Xie J, Yu S, Shao J, Jia L (March 2016). "In vitro and in vivo efficacy and safety evaluation of metapristone and mifepristone as cancer metastatic chemopreventive agents". Biomed. Pharmacother. 78: 291–300. doi:10.1016/j.biopha.2016.01.017. PMID 26898454.
- Chen W, Xiao Y, Chen J, Liu J, Shao J, Li T, Zhu Y, Ma J, Gao Y, Wang J, Xu J, Lu Y, Jia L (December 2017). "Sex-related pharmacokinetic differences and mechanisms of metapristone (RU486 metabolite)". Sci Rep. 7 (1): 17190. Bibcode:2017NatSR...717190C. doi:10.1038/s41598-017-17225-0. PMC 5719405. PMID 29215040.
- United States Pharmacopeial Convention (2006). USP DI: United States Pharmacopeia Dispensing Information. United States Pharmacopeial Convention. p. 1992. ISBN 978-1-56363-574-8.
- Heikinheimo O, Kekkonen R, Lähteenmäki P (December 2003). "The pharmacokinetics of mifepristone in humans reveal insights into differential mechanisms of antiprogestin action". Contraception. 68 (6): 421–6. doi:10.1016/S0010-7824(03)00077-5. PMID 14698071.