Betaxolol
 | |
 | |
| Names | |
|---|---|
| Pronunciation | be tax' oh lol[1] |
| Trade names | Kerlone[1] |
IUPAC name
| |
| Clinical data | |
| Drug class | Cardioselective beta blocker[1] |
| Main uses | High blood pressure, glaucoma[1] |
| Side effects | By mouth: Slow heart rate, low blood pressure, tiredness, dizziness, depression, trouble sleeping, memory loss, sexual dysfunction[1] Eye drop: Eye irritation[2] |
| WHO AWaRe | UnlinkedWikibase error: ⧼unlinkedwikibase-error-statements-entity-not-set⧽ |
| Pregnancy category |
|
| Routes of use | By mouth, eye drop |
| Typical dose | 10 to 40 mg OD[1] |
| External links | |
| AHFS/Drugs.com | Systemic: Monograph Eye: Monograph |
| MedlinePlus | a609023 |
| Legal | |
| Legal status |
|
| Pharmacokinetics | |
| Bioavailability | 89% |
| Metabolism | Liver |
| Elimination half-life | 14–22 hours |
| Excretion | Kidney (20%) |
| Chemical and physical data | |
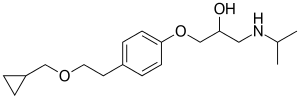
| Formula | C18H29NO3 |
| Molar mass | 307.434 g·mol−1 |
| 3D model (JSmol) | |
| Chirality | Racemic mixture |
SMILES
| |
InChI
| |
Betaxolol, sold under the brand name Kerlone among others, is a medication used to treat glaucoma and high blood pressure.[1] It reduces eye pressure by about 30%.[3] It is not a first line treatment for blood pressure.[4] It is take by mouth or used as an eye drop.[1]
Common side effects by mouth include slow heart rate, low blood pressure, tiredness, dizziness, depression, trouble sleeping, memory loss, and sexual dysfunction.[1] At high doses it can induce bronchospasm.[1] Common side effects from the eye drops include eye irritation.[2] Safety in pregnancy is unclear.[4] It is a selective beta1 receptor blocker.[1] As an eye drop it is thought to work by reducing the production of aqueous humor within the eye.[3]
Betaxolol was patented in 1975 and approved for medical use in 1983.[5] It is available as a generic medication.[1] In the United Kingdom a 5 ml bottle of eye drops costs the NHS about £2 as of 2021.[2] In the United States this amount costs about 21 USD.[6]
Medical uses
- Oral: for the management of hypertension
- Ophthalmic: for the management of glaucoma
- the drug seems to have an effect of neuroprotection in glaucoma treatment
Dosage
By mouth it is usually started at a dose of 10 mg once per day, which may be increased up to 40 mg per day.[1]
The eye drops are applied twice per day.[2]
Contraindications
- Hypersensitivity to the drug
- Patients with sinus bradycardia, heart block greater than first degree, cardiogenic shock, and overt cardiac failure
Side effects
History
Betaxolol was approved by the U.S. Food and Drug Administration (FDA) for ocular use as a 0.5% solution (Betoptic) in 1985 and as a 0.25% solution (Betoptic S) in 1989.
Brand names
Brand names include Betoptic, Betoptic S, Lokren, Kerlone.
See also
References
- 1 2 3 4 5 6 7 8 9 10 11 12 13 "Betaxolol". LiverTox: Clinical and Research Information on Drug-Induced Liver Injury. National Institute of Diabetes and Digestive and Kidney Diseases. 2012. Archived from the original on 15 May 2021. Retrieved 10 January 2022.
- 1 2 3 4 BNF 81: March-September 2021. BMJ Group and the Pharmaceutical Press. 2021. p. 1224. ISBN 978-0857114105.
- 1 2 "Betaxolol (EENT) Monograph for Professionals". Drugs.com. Archived from the original on 24 January 2021. Retrieved 10 January 2022.
- 1 2 "Betaxolol (Systemic) Monograph for Professionals". Drugs.com. Archived from the original on 6 August 2021. Retrieved 10 January 2022.
- ↑ Fischer J, Ganellin CR (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 461. ISBN 9783527607495. Archived from the original on 2017-09-08. Retrieved 2021-03-21.
- ↑ "Betaxolol Prices, Coupons & Savings Tips - GoodRx". GoodRx. Archived from the original on 6 October 2016. Retrieved 10 January 2022.
External links
| Identifiers: |
|---|