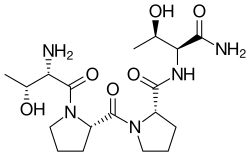
Rapastinel
Rapastinel (INN) (former developmental code name GLYX-13) is a novel antidepressant that was under development by Allergan (previously Naurex) as an adjunctive therapy for the treatment of treatment-resistant depression.[1][2] It is a centrally active, intravenously administered (non-orally active) amidated tetrapeptide that acts as a novel and selective modulator of the NMDA receptor.[1][2][3] The drug is a rapid-acting and long-lasting antidepressant as well as robust cognitive enhancer by virtue of its ability to enhance NMDA receptor-mediated signal transduction and synaptic plasticity.[1][2][3]
 | |
 | |
| Clinical data | |
|---|---|
| Other names | GLYX-13; BV-102 |
| Routes of administration | Intravenous |
| ATC code |
|
| Legal status | |
| Legal status | |
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| Chemical and physical data | |
| Formula | C18H31N5O6 |
| Molar mass | 413.475 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
On March 3, 2014, the U.S. FDA granted Fast Track designation to the development of rapastinel as an adjunctive therapy in treatment-resistant major depressive disorder.[4] As of 2015, the drug had completed phase II clinical development for this indication and achieved proof of concept as a rapid-acting antidepressant by demonstrating reduced depressive symptoms at days 1 through 7, as assessed by the HAM-D, without eliciting psychotomimetic or other significant side effects.[5] On January 29, 2016, Allergan (who acquired Naurex in July 2015) announced that rapastinel had received Breakthrough Therapy designation from the U.S. FDA for adjunctive treatment of major depressive disorder.[6]
Rapastinel was originally invented by Joseph Moskal, the co-founder of Naurex, via structural modification of B6B21, a monoclonal antibody that similarly binds to and modulates the NMDA receptor.[2][7][8][9] Rapastinel binds to a novel and unique domain on the NMDA receptor complex that is distinct from the glycine co-agonist binding site.[3][10] Rapastinel exhibits a biphasic dose response in vitro.[3][11] At therapeutically relevant concentrations, rapastinel enhances glutamate-mediated NMDA receptor activity, independent of glycine co-agonism, and enhances the magnitude of NMDAR-mediated synaptic plasticity at excitatory synapses in the mPFC.[3][11] Positive modulation of NMDA receptors by rapastinel produces antidepressant effects that are convergent with the NMDA receptor antagonist ketamine, however, rapastinel has no ketamine-like side effects such as cognitive impairment and psychotomimetic symptoms.[12][13]
In addition to its rapid and sustained antidepressant effects, rapastinel has been shown to enhance memory and learning in both young adult and learning-impaired, aging rat models.[14] It has been shown to increase Schaffer collateral-CA1 long-term potentiation in vitro. In concert with a learning task, rapastinel has also been shown to elevate gene expression of hippocampal NR1, a subunit of the NMDA receptor, in three-month-old rats.[15] Neuroprotective effects have also been demonstrated in Mongolian Gerbils by delaying the death of CA1, CA3, and dentate gyrus pyramidal neurons under glucose and oxygen-deprived conditions.[16]
On March 6, 2019, Allergan announced rapastinel failed to differentiate from placebo during phase III trials.[17] Early successful clinical studies of rapastinel in depression spurred the development of next-generation compounds with similar mechanisms of action including apimostinel (GATE-202, NRX-1074), a 2nd generation analog with improved potency, and zelquistinel (GATE-251, AGN-241751), a 3rd generation small molecule with improved potency and high oral bioavailability.[18]
References
- Henter, Ioline D.; Park, Lawrence T.; Zarate, Carlos A. (May 2021). "Novel Glutamatergic Modulators for the Treatment of Mood Disorders: Current Status". CNS Drugs. 35 (5): 527–543. doi:10.1007/s40263-021-00816-x. ISSN 1179-1934. PMC 8201267. PMID 33904154.
- Moskal JR, Burgdorf JS, Stanton PK, Kroes RA, Disterhoft JF, Burch RM, Amin Khan M (2016). "The Development of Rapastinel (Formerly GLYX-13); a rapid acting and long lasting antidepressant". Curr Neuropharmacol. 15 (1): 47–56. doi:10.2174/1570159x14666160321122703. PMC 5327451. PMID 26997507.
- Donello, John E.; Banerjee, Pradeep; Li, Yong-Xin; Guo, Yuan-Xing; Yoshitake, Takashi; Zhang, Xiao-Lei; Miry, Omid; Kehr, Jan; Stanton, Patric K.; Gross, Amanda L.; Burgdorf, Jeffery S. (2019-03-01). "Positive N-Methyl-D-Aspartate Receptor Modulation by Rapastinel Promotes Rapid and Sustained Antidepressant-Like Effects". The International Journal of Neuropsychopharmacology. 22 (3): 247–259. doi:10.1093/ijnp/pyy101. ISSN 1469-5111. PMC 6403082. PMID 30544218.
- FDA Grants Fast Track Designation to Naurex's Rapid-Acting Novel Antidepressant GLYX-13 http://www.prnewswire.com/news-releases/fda-grants-fast-track-designation-to-naurexs-rapid-acting-novel-antidepressant-glyx-13-248174561.html
- Preskorn, Sheldon; Macaluso, Matthew; Mehra, D. O. Vishaal; Zammit, Gary; Moskal, Joseph R.; Burch, Ronald M.; GLYX-13 Clinical Study Group (March 2015). "Randomized proof of concept trial of GLYX-13, an N-methyl-D-aspartate receptor glycine site partial agonist, in major depressive disorder nonresponsive to a previous antidepressant agent". Journal of Psychiatric Practice. 21 (2): 140–149. doi:10.1097/01.pra.0000462606.17725.93. ISSN 1538-1145. PMID 25782764. S2CID 36800194.
- plc, Allergan. "Allergan's Rapastinel Receives FDA Breakthrough Therapy Designation for Adjunctive Treatment of Major Depressive Disorder (MDD)". www.prnewswire.com. Retrieved 2022-05-13.
- Haring R, Stanton PK, Scheideler MA, Moskal JR (1991). "Glycine-like modulation of N-methyl-D-aspartate receptors by a monoclonal antibody that enhances long-term potentiation". J. Neurochem. 57 (1): 323–32. doi:10.1111/j.1471-4159.1991.tb02131.x. PMID 1828831. S2CID 37941813.
- Moskal JR, Kuo AG, Weiss C, Wood PL, O'Connor Hanson A, Kelso S, Harris RB, Disterhoft JF (2005). "GLYX-13: a monoclonal antibody-derived peptide that acts as an N-methyl-D-aspartate receptor modulator". Neuropharmacology. 49 (7): 1077–87. doi:10.1016/j.neuropharm.2005.06.006. PMID 16051282. S2CID 7372648.
- Burgdorf, Jeffrey; Zhang, Xiao-lei; Weiss, Craig; Matthews, Elizabeth; Disterhoft, John F.; Stanton, Patric K.; Moskal, Joseph R. (2011). "The N-methyl-d-aspartate receptor modulator GLYX-13 enhances learning and memory, in young adult and learning impaired aging rats". Neurobiology of Aging. 32 (4): 698–706. doi:10.1016/j.neurobiolaging.2009.04.012. ISSN 0197-4580. PMC 3035742. PMID 19446371.
- Pothula, Santosh; Liu, Rong-Jian; Wu, Min; Sliby, Alexa-Nicole; Picciotto, Marina R.; Banerjee, Pradeep; Duman, Ronald S. (March 2021). "Positive modulation of NMDA receptors by AGN-241751 exerts rapid antidepressant-like effects via excitatory neurons". Neuropsychopharmacology. 46 (4): 799–808. doi:10.1038/s41386-020-00882-7. ISSN 1740-634X. PMC 8027594. PMID 33059355.
- Zhang, Xiao-lei; Sullivan, John A.; Moskal, Joseph R.; Stanton, Patric K. (December 2008). "A NMDA receptor glycine site partial agonist, GLYX-13, simultaneously enhances LTP and reduces LTD at Schaffer collateral-CA1 synapses in hippocampus". Neuropharmacology. 55 (7): 1238–1250. doi:10.1016/j.neuropharm.2008.08.018. ISSN 0028-3908. PMC 2661239. PMID 18796308.
- Pothula, Santosh; Kato, Taro; Liu, Rong-Jian; Wu, Min; Gerhard, Danielle; Shinohara, Ryota; Sliby, Alexa-Nicole; Chowdhury, Golam M. I.; Behar, Kevin L.; Sanacora, Gerard; Banerjee, Pradeep (September 2021). "Cell-type specific modulation of NMDA receptors triggers antidepressant actions". Molecular Psychiatry. 26 (9): 5097–5111. doi:10.1038/s41380-020-0796-3. ISSN 1476-5578. PMID 32488125. S2CID 219175373.
- Kato, Taro; Duman, Ronald S. (January 2020). "Rapastinel, a novel glutamatergic agent with ketamine-like antidepressant actions: Convergent mechanisms". Pharmacology, Biochemistry, and Behavior. 188: 172827. doi:10.1016/j.pbb.2019.172827. ISSN 1873-5177. PMID 31733218. S2CID 207976034.
- Burgdorf, Jeffrey; Zhang, Xiao-lei; Weiss, Craig; Matthews, Elizabeth; Disterhoft, John F.; Stanton, Patric K.; Moskal, Joseph R. (2011). "The N-methyl-d-aspartate receptor modulator GLYX-13 enhances learning and memory, in young adult and learning impaired aging rats". Neurobiology of Aging. 32 (4): 698–706. doi:10.1016/j.neurobiolaging.2009.04.012. PMC 3035742. PMID 19446371.
- Moskal, Joseph R.; Kuo, Amy G.; Weiss, Craig; Wood, Paul L.; O'Connor Hanson, Amy; Kelso, Stephen; Harris, Robert B.; Disterhoft, John F. (2005). "GLYX-13: A monoclonal antibody-derived peptide that acts as an N-methyl-d-aspartate receptor modulator". Neuropharmacology. 49 (7): 1077–87. doi:10.1016/j.neuropharm.2005.06.006. PMID 16051282. S2CID 7372648.
- Stanton, Patric K.; Potter, Pamela E.; Aguilar, Jennifer; Decandia, Maria; Moskal, Joseph R. (2009). "Neuroprotection by a novel NMDAR functional glycine site partial agonist, GLYX-13". NeuroReport. 20 (13): 1193–7. doi:10.1097/WNR.0b013e32832f5130. PMID 19623090. S2CID 6269255.
- "Allergan Announces Phase 3 Results for Rapastinel as an Adjunctive Treatment of Major Depressive Disorder (MDD)".
- "Home - Gate Neurosciences". Retrieved 2022-05-13.
External links
- Rapastinel - AdisInsight
- Rapastinel (GLYX-13) - Naurex, Inc.
- Rapastinel Receives FDA Breakthrough Therapy Designation - Allergan plc. press release