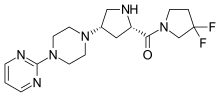
Gosogliptin
 | |
| Clinical data | |
|---|---|
| Other names | PF-734200 |
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C17H24F2N6O |
| Molar mass | 366.417 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
Gosogliptin (INN; trade name Saterex) is a drug for the treatment of type II diabetes. It is in the class of dipeptidyl peptidase-4 (DPP-4) inhibitors (also called gliptins). It was discovered[1] and developed through Phase 1[2][3][4] and Phase 2[5][6][7] by Pfizer. The crystal structure of DPP-4 in complex with gosogliptin is available.[8] Its metabolism, excretion and pharmacokinetics in rat, dog and human have been described.[9] A cost efficient route has been published.[10] Other studies[11] including Phase 3 studies were conducted in Russia.[12][13] It is approved for use in Russia.[14]
References
- ↑ Ammirati MJ, Andrews KM, Boyer DD, Brodeur AM, Danley DE, Doran SD, Hulin B, Liu S, McPherson RK, Orena SJ, Parker JC, Polivkova J, Qiu X, Soglia CB, Treadway JL, VanVolkenburg MA, Wilder DC, Piotrowski DW (April 2009). "(3,3-Difluoro-pyrrolidin-1-yl)-[(2S,4S)-(4-(4-pyrimidin-2-yl-piperazin-1-yl)-pyrrolidin-2-yl]-methanone: a potent, selective, orally active dipeptidyl peptidase IV inhibitor". Bioorganic & Medicinal Chemistry Letters. 19 (7): 1991–1995. doi:10.1016/j.bmcl.2009.02.041. PMID 19275964.
- ↑ Dai H, Gustavson SM, Preston GM, Eskra JD, Calle R, Hirshberg B (2008). "Non-linear increase in GLP-1 levels in response to DPP-IV inhibition in healthy adult subjects". Diabetes, Obesity and Metabolism. 10 (6): 506–513. doi:10.1111/j.1463-1326.2007.00742.x. PMID 18284437. S2CID 13417601.
- ↑ Dai H, Johnson SL, Terra SG, Marbury TC, Smith WB, Alcorn H, Boyd RA, Wang R, Nguyen TT (July 2011). "The pharmacokinetics of PF-734200, a DPP-IV inhibitor, in subjects with renal insufficiency". British Journal of Clinical Pharmacology. 72 (1): 85–91. doi:10.1111/j.1365-2125.2011.03954.x. PMC 3141189. PMID 21366665.
- ↑ Muto C, Dai H, Teeter JG, Johnson S, Cropp AB, Chiba K, Suwa T (2012). "The pharmacokinetics and pharmacodynamics of PF-00734200, a DPP-IV inhibitor, in healthy Japanese subjects". International Journal of Clinical Pharmacology and Therapeutics. 50 (7): 505–509. doi:10.5414/CP201614. PMID 22541754.
- ↑ Rosenstock J, Lewin AJ, Norwood P, Somayaji V, Nguyen TT, Teeter JG, Johnson SL, Dai H, Terra SG (2011). "Efficacy and safety of the dipeptidyl peptidase‐4 inhibitor PF‐734200 added to metformin in Type 2 diabetes". Diabetic Medicine. 28 (4): 464–469. doi:10.1111/j.1464-5491.2010.03181.x. PMID 21392067. S2CID 205550845.
- ↑ "A Phase 2, Randomized, Double-Blind, Placebo-Controlled, Parallel Group Study to Evaluate the Efficacy and Safety of 12-Week Administration of PF-00734200 to Subjects with Type 2 Diabetes Mellitus and Insufficient Glycemic Control on Metformin Treatment". 9 June 2011.
{{cite journal}}: Cite journal requires|journal=(help) - ↑ "A Phase 2a, Randomized, Placebo-Controlled, Parallel Group, Multiple-Dose Study to Evaluate the Efficacy, Safety, and Tolerability of 12-Week Oral Administration of PF-00734200 Tablets to Subjects with Type 2 Diabetes Mellitus on Stable Treatment with Metformin". 22 June 2010.
{{cite journal}}: Cite journal requires|journal=(help) - ↑ "RCSB PDB - 3F8S: Crystal structure of dipeptidyl peptidase IV in complex with inhibitor".
- ↑ Sharma R, Sun H, Piotrowski DW, Ryder TF, Doran SD, Dai H, Prakash C (November 2012). "Metabolism, Excretion, and Pharmacokinetics of ((3,3-Difluoropyrrolidin-1-yl)((2 S ,4 S )-4-(4-(pyrimidin-2-yl)piperazin-1-yl)pyrrolidin-2-yl)methanone, a Dipeptidyl Peptidase Inhibitor, in Rat, Dog and Human". Drug Metabolism and Disposition. 40 (11): 2143–2161. doi:10.1124/dmd.112.047316. ISSN 0090-9556. PMID 22896728. S2CID 14277073.
- ↑ Lafrance D, Caron S (January 2012). "New Synthetic Route to a Dipeptidyl Peptidase-4 Inhibitor". Organic Process Research & Development. 16 (3): 409–14. doi:10.1021/op200309z.
- ↑ Nedosugova LV, Petunina NA, Galstyan KO (2014). "Initial investigation of efficacy and safety of a new dipeptidyl peptidase-4 inhibitor, gosogliptin, for type 2 diabetes in Russia". Diabetes Mellitus. 17 (4): 81–86. doi:10.14341/DM2014481-86.
- ↑ Galstyan KO, Nedosugova LV, Petunina NA, Trakhtenberg JA, Vostokova NV, Karavaeva OV, Chasovskaya TE (April 2016). "Comparison of the novel Russian DPP-4 inhibitor gosogliptin with vildagliptin in patients with type 2 diabetes mellitus". Diabetes Mellitus. 19 (1): 89‐96. doi:10.14341/DM7233.
- ↑ "Evaluate Efficacy and Safety of Gosogliptin as Monotherapy and in Combination with Metformin vs. Vildagliptin as Monotherapy and in Combination with Metformin in Drug-naive Type 2 Diabetic Patients". 18 March 2017.
{{cite journal}}: Cite journal requires|journal=(help) - ↑ "SatRx LLC Announces First Registration in Russia of SatRx (gosogliptin), an Innovative Drug for Treatment of Type 2 Diabetes" (Press release). SatRx LLC.
This article is issued from Offline. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.