Selexipag
Selexipag, sold under the brand name Uptravi, is a medication developed by Actelion for the treatment of pulmonary arterial hypertension (PAH).[1] Selexipag and its active metabolite, ACT-333679 (or MRE-269, the free carboxylic acid), are agonists of the prostacyclin receptor, which leads to vasodilation in the pulmonary circulation.[2] It is taken by mouth or administered intravenously.[1][3]
 | |
| Clinical data | |
|---|---|
| Trade names | Uptravi |
| Other names | ACT-293987, NS-304 |
| License data | |
| Routes of administration | By mouth, intravenous |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 49% |
| Protein binding | 99% |
| Metabolism | Activation by carboxylesterases, inactivation by CYP2C8 and others |
| Metabolites | ACT-333679, the free acid (active metabolite) |
| Elimination half-life | 0.8–2.5 h (selexipag) and 6.2–13.5 h (ACT-333679) |
| Excretion | 93% faeces |
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| ECHA InfoCard | 100.237.916 |
| Chemical and physical data | |
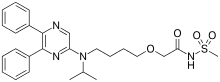
| Formula | C26H32N4O4S |
| Molar mass | 496.63 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
| | |
Contraindications
In Europe, use of selexipag together with strong inhibitors of the liver enzyme CYP2C8, such as gemfibrozil, is contraindicated because it increases concentrations of selexipag twofold, and its active metabolite 11-fold, potentially leading to more adverse effects.[4]
Adverse effects
The adverse effects of selexipag are similar to those of intravenous prostacyclins used for pulmonary arterial hypertension. Common side effects include headache and jaw pain. An increased risk for hyperthyroidism has also been noted in people taking selexipag.[5]
Pharmacology
Mechanism of action
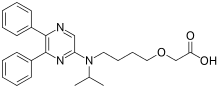
Selexipag and its active metabolite ACT-333679 act on the prostacyclin receptor of lung tissue, with the latter being 37-fold more potent. They are selective for the prostacyclin receptor. Binding to this receptor leads to three major effects: increased vasodilation of the arteries, decreased cell proliferation and inhibition of platelet aggregation,[5] all beneficial in the treatment of pulmonary arterial hypertension.
Pharmacokinetics
Selexipag is quickly absorbed from the gut and hydrolyzed in the intestines and the liver to ACT-333679 by carboxylesterases. Absolute bioavailability is about 49%, most likely because of a high first-pass effect. Highest concentrations in the blood plasma are reached after one to three hours for selexipag and after three to four hours for the active metabolite. When in the circulation, about 99% of both substances are bound to plasma proteins, namely to albumin and alpha-1-acid glycoprotein to equal amounts.[5]
The liver enzymes CYP2C8 and, to a lesser extent, CYP3A4, hydroxylate and dealkylate the active substance, thereby inactivating it. Besides, ACT-333679 is glucuronidized by the enzymes UGT1A3 and UGT2B7. The terminal half-life of selexipag is 0.8 to 2.5 hours, that of the active metabolite is 6.2 to 13.5 hours.[5]
History
The U.S. Food and Drug Administration (FDA) granted selexipag orphan drug status for PAH.[6] It was approved by the FDA on 22 December 2015.[6]
In Europe, the drug was approved in May 2016.[5]
Society and culture
Economics
The expected price for the drug in the US is $160,000 to $170,000 per patient before rebates.[7]
References
- "Uptravi- selexipag tablet, coated Uptravi Titration Pack- selexipag kit". DailyMed. Retrieved 30 July 2021.
- Sitbon O, Morrell N (December 2012). "Pathways in pulmonary arterial hypertension: the future is here". European Respiratory Review. 21 (126): 321–7. doi:10.1183/09059180.00004812. PMID 23204120.
- "Uptravi (selexipag) Receives FDA Approval for Intravenous Use in Adult Patients with Pulmonary Arterial Hypertension (PAH)". Janssen Pharmaceutical Companies (Press release). 30 July 2021. Retrieved 30 July 2021.
- Information des Bundesamtes für Sicherheit im Gesundheitswesen zu Uptravi (in German), Österreichisches Bundesamt für Sicherheit im Gesundheitswesen, 2017-06-07
- "Uptravi: Authorisation details". European Medicines Agency. 2016-05-12.
- New Drug Approved for Rare Lung Disorder. PPN. 23 Dec 2015 Has link to GRIPHON study results
- "Actelion sees Uptravi price of $160,000-170,000/patient". Reuters. 2016-01-05. Retrieved 2016-01-06.
External links
- "Selexipag". Drug Information Portal. U.S. National Library of Medicine.
- Clinical trial number NCT03187678 for "Safety Study of the Switch From Oral Selexipag to Intravenous Selexipag in Subjects With Stable Pulmonary Arterial Hypertension" at ClinicalTrials.gov