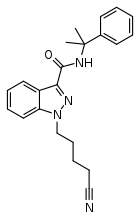
CUMYL-4CN-BINACA
 | |
| Legal status | |
|---|---|
| Legal status |
|
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C22H24N4O |
| Molar mass | 360.461 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
CUMYL-4CN-BINACA (also known as CUMYL-CYBINACA or SGT-78) is an indazole-3-carboxamide based synthetic cannabinoid that has been sold online as a designer drug.[1][2][3][4][5][6] It is a potent agonist for cannabinoid receptors CB1 and CB2, with in vitro EC50 values of 0.58 nM and 6.12 nM, respectively.[7] In mice, CUMYL-4CN-BINACA produces hypothermic and pro-convulsant effects via the CB1 receptor,[7] and anecdotal reports suggest it has an active dose of around 0.1 mg in humans.[8]
CUMYL-4CN-BINACA is metabolized to produce cyanide, raising concerns about liver toxicity.[6] There is one reported case of hyperthermia, rhabdomyolysis, and kidney failure associated with its use.[9]
See also
- 4CN-AMB-BUTINACA
- 5F-CUMYL-PINACA
- 5F-SDB-006
- ADAMANTYL-THPINACA
- CUMYL-PICA
- CUMYL-PINACA
- CUMYL-THPINACA
- SDB-006
- NNE1
References
- ↑ "4-cyano CUMYL-BUTINACA". Cayman Chemical.
- ↑ Yeter O (November 2017). "Identification of the Synthetic Cannabinoid 1-(4-cyanobutyl)-N-(2-phenylpropan-2-yl)-1H-indazole-3-carboxamide (CUMYL-4CN-BINACA) in Plant Material and Quantification in Post-Mortem Blood Samples". Journal of Analytical Toxicology. 41 (9): 720–728. doi:10.1002/dta.2248. PMID 28977413.
- ↑ Staeheli SN, Poetzsch M, Veloso VP, Bovens M, Bissig C, Steuer AE, Kraemer T (January 2018). "In vitro metabolism of the synthetic cannabinoids CUMYL-PINACA, 5F-CUMYL-PINACA, CUMYL-4CN-BINACA, 5F-CUMYL-P7AICA and CUMYL-4CN-B7AICA". Drug Testing and Analysis. 10 (1): 148–157. doi:10.1002/dta.2298. PMID 28885775.
- ↑ Bovens M, Bissig C, Staeheli SN, Poetzsch M, Pfeiffer B, Kraemer T (December 2017). "Structural characterization of the new synthetic cannabinoids CUMYL-PINACA, 5F-CUMYL-PINACA, CUMYL-4CN-BINACA, 5F-CUMYL-P7AICA and CUMYL-4CN-B7AICA". Forensic Science International. 281: 98–105. doi:10.1016/j.forsciint.2017.10.020. PMID 29125990.
- ↑ Ölmez, Nevin Arıkan; Kapucu, Hicran; Altun, Neslihan Çallı; Eren, Bülent (January 2018). "Identification of the synthetic cannabinoid N-(2-phenyl-propan-2-yl)-1-(4-cyanobutyl)-1H-indazole-3-carboxamide (CUMYL-4CN-BINACA) in a herbal mixture product". Forensic Toxicology. 36 (1): 192–199. doi:10.1007/s11419-017-0372-y. ISSN 1860-8965. S2CID 28058750.
- 1 2 Åstrand, Anna; Vikingsson, Svante; Lindstedt, Daniel; Thelander, Gunilla; Gréen, Henrik; Kronstrand, Robert; Wohlfarth, Ariane (2018). "Metabolism study for CUMYL-4CN-BINACA in human hepatocytes and authentic urine specimens: Free cyanide is formed during the main metabolic pathway". Drug Testing and Analysis. 10 (8): 1270–1279. doi:10.1002/dta.2373. ISSN 1942-7611. PMID 29577658.
- 1 2 Kevin, Richard; Anderson, Lyndsey; McGregor, Iain; Boyd, Rochelle; Manning, Jamie; Glass, Michelle; Connor, Mark; Banister, Samuel (2019). "CUMYL-4CN-BINACA is an efficacious and potent pro-convulsant synthetic cannabinoid receptor agonist". Frontiers in Pharmacology. 10: 595. doi:10.3389/fphar.2019.00595. PMC 6549035. PMID 31191320.
- ↑ "EMCDDA–Europol Joint Report on a new psychoactive substance: 1-(4-cyanobutyl)-N-(2-phenylpropan-2-yl)indazole-3-carboxamide (CUMYL-4CN-BINACA)". www.emcdda.europa.eu.
- ↑ El Zahran, Tharwat (August 2018). "A novel synthetic cannabinoid (Cumyl-4-cyano-BINACA) resulting in hyperthermia, rhabdomyolysis, and renal failure in a 29-year-old patient: it's not meningitis". Clinical Toxicology. 57 (6): 421–422. doi:10.1080/15563650.2018.1534241. PMID 30442067. S2CID 53566116.
This article is issued from Offline. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.