Cannabinor
 | |
| Identifiers | |
|---|---|
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| Chemical and physical data | |
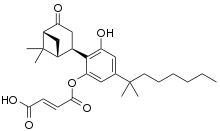
| Formula | C28H36O6 |
| Molar mass | 468.590 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
Cannabinor (PRS-211,375) is a drug which acts as a potent and selective cannabinoid CB2 receptor agonist.[1][2] It is classed as a "nonclassical" cannabinoid with a chemical structure similar to that of cannabidiol. It has a CB2 affinity of 17.4nM vs 5585nM at CB1, giving it over 300x selectivity for CB2.[3] It showed analgesic effects in animal studies especially in models of neuropathic pain, but failed in Phase IIb human clinical trials due to lack of efficacy.[4]
See also
References
- ↑ Gratzke C, Streng T, Stief CG, Downs TR, Alroy I, Rosenbaum JS, Andersson KE, Hedlund P (June 2010). "Effects of cannabinor, a novel selective cannabinoid 2 receptor agonist, on bladder function in normal rats". European Urology. 57 (6): 1093–100. doi:10.1016/j.eururo.2010.02.027. PMID 20207474.
- ↑ Gratzke C, Streng T, Stief CG, Alroy I, Limberg BJ, Downs TR, et al. (February 2011). "Cannabinor, a selective cannabinoid-2 receptor agonist, improves bladder emptying in rats with partial urethral obstruction". The Journal of Urology. 185 (2): 731–6. doi:10.1016/j.juro.2010.09.080. PMID 21168864.
- ↑ Bow EW, Rimoldi JM (2016). "The Structure-Function Relationships of Classical Cannabinoids: CB1/CB2 Modulation". Perspectives in Medicinal Chemistry. 8: 17–39. doi:10.4137/PMC.S32171. PMC 4927043. PMID 27398024.
- ↑ Nevalainen T (2013). "Recent development of CB2 selective and peripheral CB1/CB2 cannabinoid receptor ligands". Current Medicinal Chemistry. 21 (2): 187–203. doi:10.2174/09298673113206660296. PMID 24164198.
This article is issued from Offline. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.