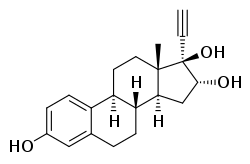
Ethinylestriol
 | |
| Clinical data | |
|---|---|
| Other names | EE3; 17α-Ethynylestriol; 17α-Ethynylestra-1,3,5(10)-triene-3,16α,17β-triol |
| Routes of administration | By mouth |
| Drug class | Estrogen |
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C20H24O3 |
| Molar mass | 312.409 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
Ethinylestriol (EE3), or 17α-ethynylestriol, also known as 17α-ethynylestra-1,3,5(10)-triene-3,16α,17β-triol, is a synthetic estrogen which was never marketed.[1] Nilestriol, the 3-cyclopentyl ether of ethinylestriol, is a prodrug of ethinylestriol, and is a more potent estrogen in comparison,[1] but, in contrast to ethinylestriol, has been marketed.[2] Ethinylestriol has been found to reduce the risk of 7,12-dimethylbenz(a)anthracene (DMBA)-induced mammary cancer when given as a prophylactic in animal models, while other estrogens like ethinylestradiol and diethylstilbestrol were ineffective.[3]
See also
References
- 1 2 McGuire W (14 December 2013). Experimental Biology. Springer Science & Business Media. pp. 161–. ISBN 978-1-4757-4673-0.
- ↑ Elks J (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 891–. ISBN 978-1-4757-2085-3.
- ↑ Lemon HM (December 1987). "Antimammary carcinogenic activity of 17-alpha-ethinyl estriol". Cancer. 60 (12): 2873–81. doi:10.1002/1097-0142(19871215)60:12<2873::AID-CNCR2820601204>3.0.CO;2-B. PMID 3119187.
This article is issued from Offline. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.