Nemifitide
 | |
| Clinical data | |
|---|---|
| Routes of administration | Subcutaneous injection |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Elimination half-life | 15-30 minutes[1] |
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| Chemical and physical data | |
| Formula | C33H43FN10O6 |
| Molar mass | 694.769 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
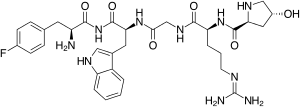
Nemifitide (INN-00835) is a novel antidepressant drug with a pentapeptide structure similar to that of melanocyte-inhibiting factor (MIF-1) and the amino acid sequence 4-F-Phe-4-OH-Pro-Arg-Gly-Trp-NH2.[1][2] It is under development by Tetragenex (previously Innapharma, Inc.) for the treatment of major depressive disorder.[3] It has been given to over 430 people over the course of 12 clinical trials throughout a little over the past decade and has reached Phase III studies, but has not yet been approved for marketing in any country.[3]
Nemifitide has shown mixed efficacy in alleviating depressive symptoms,[4][5] but in the cases in which it has worked it has proven to have a rapid onset of action (~5–7 days), few to no side effects, and an excellent safety profile.[5] However, it is inactive orally and must be administered via subcutaneous injection. Remarkably, despite having a very short half-life of only 15–30 minutes,[1] in most or all studies assessing its efficacy nemifitide has been administered merely once daily via the subcutaneous route and yet is effective for depression.
The mechanism of action of nemifitide is unclear, but since MIF-1 has been demonstrated to have similar antidepressant effects[6] it may act in an analogous manner.[1][2] Possibly of interest however is that nemifitide binds to several receptors including 5-HT2A (where it has been shown to act as an antagonist), NPY1, bombesin, and MC4 and MC5, though at only micromolar concentrations.[5] Whether any of these relatively weak actions are of any clinical significance is unclear.
See also
- Melanocyte-inhibiting factor
References
- 1 2 3 4 Foye WO, Lemke TL, Williams DA (1 September 2007). Foye's principles of medicinal chemistry. Lippincott Williams & Wilkins. p. 596. ISBN 978-0-7817-6879-5. Retrieved 15 July 2016.
- 1 2 Holtzheimer PE, Nemeroff CB (December 2008). "Novel targets for antidepressant therapies". Current Psychiatry Reports. 10 (6): 465–73. doi:10.1007/s11920-008-0075-5. PMC 4413474. PMID 18980729.
- 1 2 "Tetragenex Pharmaceuticals Inc. - Product Technology - Nemifitide". Archived from the original on 25 March 2012. Retrieved 13 June 2011.
- ↑ Rakofsky JJ, Holtzheimer PE, Nemeroff CB (June 2009). "Emerging targets for antidepressant therapies". Current Opinion in Chemical Biology. 13 (3): 291–302. doi:10.1016/j.cbpa.2009.04.617. PMC 4410714. PMID 19501541.
- 1 2 3 Renato D. Alarcón (2004). Antidepressants: past, present, and future. Springer. p. 575. ISBN 978-3-540-43054-4.
- ↑ van der Velde CD (1983). "Rapid clinical effectiveness of MIF-I in the treatment of major depressive illness". Peptides. 4 (3): 297–300. doi:10.1016/0196-9781(83)90136-5. PMID 6138756. S2CID 54412069.
Further reading
- Dingemanse J (July 2003). "Nemifitide. Innapharma". Current Opinion in Investigational Drugs. 4 (7): 859–62. PMID 14619408.