Milnacipran
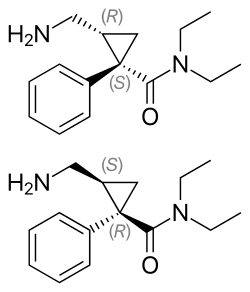 | |
 Top: (1S,2R)-milnacipran (L-milnacipran) Bottom: (1R,2S)-milnacipran (D-milnacipran) | |
| Names | |
|---|---|
| Trade names | Ixel, Joncia, Savella, Dalcipran, others |
| Other names | Milnacipran hydrochloride |
IUPAC name
| |
| Clinical data | |
| Drug class | Serotonin–norepinephrine reuptake inhibitor (SNRI)[1] |
| Main uses | Fibromyalgia, major depressive disorder[1] |
| Side effects | Nausea, headache, trouble sleeping, increased sweating, palpitations[1] |
| WHO AWaRe | UnlinkedWikibase error: ⧼unlinkedwikibase-error-statements-entity-not-set⧽ |
| Pregnancy category |
|
| Routes of use | By mouth (tablets), capsules) |
| Typical dose | 50 mg BID[1] |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a609016 |
| Legal | |
| Legal status | |
| Pharmacokinetics | |
| Bioavailability | 85% |
| Protein binding | 13% |
| Metabolism | Liver |
| Elimination half-life | 8 hours |
| Excretion | Kidney |
| Chemical and physical data | |
| Formula | C15H22N2O |
| Molar mass | 246.354 g·mol−1 |
| 3D model (JSmol) | |
| Chirality | Racemic mixture |
SMILES
| |
InChI
| |
Milnacipran, sold under the brand name Savella among others, is a medication used to treat fibromyalgia and major depressive disorder.[1] It is taken by mouth.[1]
Common side effects include nausea, headache, trouble sleeping, increased sweating, and palpitations.[1] Other side effects may include suicide in those under 25 years old, serotonin syndrome, increased blood pressure, liver problems, bleeding, and mania.[1] Safety in pregnancy is unclear.[1] It is a serotonin–norepinephrine reuptake inhibitor (SNRI).[1]
Milnacipran was approved for medical use in the United States in 2009.[1] It was denied approval in Europe in 2009 due to questionable benefits.[2] In the United States it costs about 420 USD for a month as of 2021.[3]
Medical uses
Depression
A 2007 meta-analysis showed no distinction between milnacipran and SSRIs in efficacy or discontinuation rates, including discontinuation for side effects or lack of efficacy.[4] A 2009 meta-analysis concluded that there were no differences in efficacy, acceptability and tolerability when comparing milnacipran with other antidepressant agents.[5] However, compared with TCAs, fewer people taking milnacipran dropped out due to adverse events. As with other antidepressants, 1 to 3 weeks may elapse before significant antidepressant action becomes evident.
Fibromyalgia
During its development for fibromyalgia, milnacipran was evaluated utilizing a composite responder approach. To be considered as a responder for the composite ‘treatment of fibromyalgia’ endpoint, each patient had to show concurrent and clinically meaningful improvements in pain, physical function, and global impression of disease status. A systematic review in 2015 showed moderate relief for a minority of people with fibromyalgia. Milnacipran was associated with increased adverse events when discontinuing use of the drug.[6]
Social anxiety
There is some evidence that milnacipran may be effective for social anxiety.[7]
Dosage
It is gradually increased to 50 mg twice per day.[1]
Contraindications
Administration of milnacipran should be avoided in individuals with the following:
- Known hypersensitivity to milnacipran (absolute contraindication)
- Patients under 15 years of age (no sufficient clinical data)
- Concomitant treatment with irreversible MAO inhibitors (e.g. tranylcypromine (Parnate), phenelzine (Nardil), >10 mg selegiline) or digitalis glycosides is an absolute contraindication.
Administration of milnacipran should be done with caution in individuals with the following:
- Concomitant treatment with parenteral epinephrine, norepinephrine, with clonidine, reversible MAO-A Inhibitors (such as moclobemide, toloxatone) or 5-HT1D-agonists (e.g. triptan migraine drugs)
- Advanced renal disease (decreased dosage required)
- Hypertrophy of the prostate gland (possibly urination hesitancy induced), with hypertension and heart disease (tachycardia may be a problem) as well as with open angle glaucoma.
Milnacipran should not be used during pregnancy because it may cross the placenta barrier and no clinical data exists on harmful effects in humans and animal studies. Milnacipran is contraindicated during lactation because it is excreted in the milk, and it is not known if it is harmful to the newborn.
Side effects
The most frequently occurring adverse reactions (≥ 5% and greater than placebo) were nausea, headache, constipation, dizziness, insomnia, hot flush, hyperhydrosis, vomiting, palpitations, mouth rate increase, dry mouth, and hypertension [FDA Savella prescribing information]. Milnacipran can have a significant impact on sexual functions, including both a decrease in sexual desire and ability. Milnacipran can cause pain of the testicles in men. The incidence of cardiovascular and anticholinergic side effects was significantly lower compared to TCAs as a controlled study with over 3,300 patients revealed. Elevation of liver enzymes without signs of symptomatic liver disease has been infrequent. Mood swing to mania has also been seen and dictates termination of treatment. In psychotic patients emergence of delirium has been noticed. Milnacipran has a low incidence of sedation but improves sleep (both duration and quality) in depressed patients. In agitated patients or those with suicidal thoughts additive sedative/anxiolytic treatment is usually indicated.[8]
Interactions
- MAOIs — hyperserotonergia (serotonin syndrome), potentially lethal hypertensive crisis
- 5-HT1 receptor agonists — coronary vasoconstriction with risk of angina pectoris and myocardial infarction
- Epinephrine, norepinephrine (also in local anesthesia) — hypertensive crisis and/or possible cardiac arrhythmia
- Clonidine — antihypertensive action of clonidine may be antagonized
- Digitalis — hemodynamic actions increased
- Triptans — there have been rare postmarketing reports of hyperserotonergia (serotonin syndrome). If concomitant treatment of milnacipran with a triptan is clinically warranted, careful observation of patient is advised when starting or increasing dosages.[9]
- Alcohol — no interactions known; however, because milnacipran can cause mild elevation of liver enzymes, caution is recommended; the FDA advises against the concomitant use of alcohol and milnacipran
Pharmacology
Pharmacodynamics
Milnacipran inhibits the reuptake of serotonin and norepinephrine in an approximately 1:3 ratio, respectively. Milnacipran exerts no significant actions on H1, α1, D1, D2, and mACh receptors, nor on benzodiazepine and opioid binding sites.[10][11][12]
Recently, levomilnacipran, the levorotatory enantiomer of milnacipran, has been found to act as an inhibitor of beta-site amyloid precursor protein cleaving enzyme-1 (BACE-1), which is responsible for β-amyloid plaque formation, and hence may be a potentially useful drug in the treatment of Alzheimer's disease.[13] Other BACE-1 inhibitors, such as CTS-21166 (ASP1720), MK-8931, and AZD3293 were in clinical trials for the treatment of Alzheimer's disease,[14] but in both cases clinical trials were halted due to a lack of positive evidence of a favorable benefit to risk ratio and both were considered unlikely to return satisfactory results.
Pharmacokinetics
Milnacipran is well absorbed after oral dosing and has a bioavailability of 85%. Meals do not have an influence on the rapidity and extent of absorption. Peak plasma concentrations are reached 2 hours after oral dosing. The elimination half-life of 8 hours is not increased by liver impairment and old age, but by significant renal disease. Milnacipran is conjugated to the inactive glucuronide and excreted in the urine as unchanged drug and conjugate. Only traces of active metabolites are found.
History
Milnacipran was first approved for the treatment of major depressive episodes in France in December 1996. It is currently marketed (as Ixel) for this indication in over 45 countries worldwide including several European countries such as Austria, Bulgaria, Finland, France, Portugal, and Russia. It is also available in Japan (as Toledomin) and Mexico (as Dalcipran). Cypress Bioscience bought the exclusive rights for approval and marketing of the drug for any purpose in the United States and Canada in 2003 from the manufacturer Laboratoires Pierre Fabre.
In January 2009 the U.S. Food and Drug Administration (FDA) approved milnacipran (under the brand name Savella) only for the treatment of fibromyalgia, making it the third medication approved for this purpose in the United States. In July and November 2009, the European Medicines Agency refused marketing authorization for a milnacipran product (under the brand name Impulsor) for the treatment of fibromylagia.[15]
References
- 1 2 3 4 5 6 7 8 9 10 11 12 "Milnacipran Monograph for Professionals". Drugs.com. Archived from the original on 27 February 2021. Retrieved 18 November 2021.
- ↑ "Milnacipran Pierre Fabre Medicament". Archived from the original on 22 September 2020. Retrieved 18 November 2021.
- ↑ "Savella Prices, Coupons & Savings Tips - GoodRx". GoodRx. Archived from the original on 17 August 2021. Retrieved 18 November 2021.
- ↑ Papakostas GI, Fava M (January 2007). "A meta-analysis of clinical trials comparing milnacipran, a serotonin--norepinephrine reuptake inhibitor, with a selective serotonin reuptake inhibitor for the treatment of major depressive disorder". European Neuropsychopharmacology. 17 (1): 32–6. doi:10.1016/j.euroneuro.2006.05.001. PMID 16762534. S2CID 27679241.
- ↑ Nakagawa A, Watanabe N, Omori IM, Barbui C, Cipriani A, McGuire H, Churchill R, Furukawa TA (July 2009). Nakagawa A (ed.). "Milnacipran versus other antidepressive agents for depression". The Cochrane Database of Systematic Reviews. 8 (3): CD006529. doi:10.1002/14651858.CD006529.pub2. PMC 4164845. PMID 19588396.
- ↑ Cording M, Derry S, Phillips T, Moore RA, Wiffen PJ (October 2015). "Milnacipran for pain in fibromyalgia in adults". The Cochrane Database of Systematic Reviews (10): CD008244. doi:10.1002/14651858.CD008244.pub3. PMC 6481368. PMID 26482422.
- ↑ Higuchi T, Briley M (February 2007). "Japanese experience with milnacipran, the first serotonin and norepinephrine reuptake inhibitor in Japan". Neuropsychiatric Disease and Treatment. 3 (1): 41–58. doi:10.2147/nedt.2007.3.1.41. PMC 2654524. PMID 19300537.
- ↑ "www.accessdata.fda.gov" (PDF). Archived (PDF) from the original on 2021-04-13. Retrieved 2021-10-14.
- ↑ "SAVELLA - milnacipran hydrochloride tablet, film coated". DailyMed. National Institute of Health. Archived from the original on 2013-10-29. Retrieved 2021-10-14.
- ↑ Moret C, Charveron M, Finberg JP, Couzinier JP, Briley M (December 1985). "Biochemical profile of midalcipran (F 2207), 1-phenyl-1-diethyl-aminocarbonyl-2-aminomethyl-cyclopropane (Z) hydrochloride, a potential fourth generation antidepressant drug". Neuropharmacology. 24 (12): 1211–9. doi:10.1016/0028-3908(85)90157-1. PMID 3005901. S2CID 46629043.
- ↑ Briley M, Prost JF, Moret C (September 1996). "Preclinical pharmacology of milnacipran". International Clinical Psychopharmacology. 11 Suppl 4: 9–14. doi:10.1097/00004850-199609004-00002. PMID 8923122. S2CID 37407165.
- ↑ Puozzo C, Panconi E, Deprez D (June 2002). "Pharmacology and pharmacokinetics of milnacipran". International Clinical Psychopharmacology. 17 Suppl 1: S25-35. doi:10.1097/00004850-200206001-00004. PMID 12369608. S2CID 45279690.
- ↑ Rizvi SM, Shaikh S, Khan M, Biswas D, Hameed N, Shakil S (2014). "Fetzima (levomilnacipran), a drug for major depressive disorder as a dual inhibitor for human serotonin transporters and beta-site amyloid precursor protein cleaving enzyme-1". CNS & Neurological Disorders Drug Targets. 13 (8): 1427–31. doi:10.2174/1871527313666141023145703. PMID 25345508.
- ↑ Menting KW, Claassen JA (2014). "β-secretase inhibitor; a promising novel therapeutic drug in Alzheimer's disease". Frontiers in Aging Neuroscience. 6: 165. doi:10.3389/fnagi.2014.00165. PMC 4104928. PMID 25100992.
- ↑ European Medicines Agency. "Questions and answers on the recommendati on for the refusal of the marketing authorisation for Milnacipran Pierre Fabre Médicament/Impulsor" (PDF). European Medicines Agency. Archived (PDF) from the original on 22 February 2014. Retrieved 30 May 2013.
External links
| Identifiers: |
|---|
- "Milnacipran". Drug Information Portal. U.S. National Library of Medicine. Archived from the original on 2021-07-25. Retrieved 2021-10-14.
- "Milnacipran hydrochloride". Drug Information Portal. U.S. National Library of Medicine. Archived from the original on 2020-10-15. Retrieved 2021-10-14.