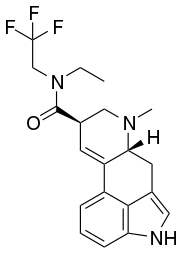
ETFELA
ETFELA (N-ethyl-N-(2,2,2-trifluoroethyl)lysergamide) is an analog of lysergic acid diethylamide (LSD) first synthesised by Jason C. Parrish as part of the research team led by David E. Nichols. In studies in vitro, it was found to be slightly more potent than LSD itself.[1][2]
 | |
| Clinical data | |
|---|---|
| Other names | LA-CH2CF3 |
| Identifiers | |
| |
| Chemical and physical data | |
| Formula | C20H22F3N3O |
| Molar mass | 377.411 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
See also
References
- Nichols DE (2012). "Structure-activity relationships of serotonin 5-HT2A agonists". Wiley Interdisciplinary Reviews: Membrane Transport and Signaling. 1 (5): 559–579. doi:10.1002/wmts.42.
- Nichols DE (2017). "Chemistry and Structure-Activity Relationships of Psychedelics". Current Topics in Behavioral Neurosciences. 36: 1–43. doi:10.1007/7854_2017_475. ISBN 978-3-662-55878-2. PMID 28401524. S2CID 4396184.
| Lysergic acid derivatives |
|
|---|---|
| Psychedelic lysergamides |
|
| Clavines | |
| Other ergolines | |
| Natural sources |
Morning glory: Argyreia nervosa (Hawaiian Baby Woodrose), Ipomoea spp.(Morning Glory, Tlitliltzin, Badoh Negro), Rivea corymbosa (Coaxihuitl, Ololiúqui) |
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.