Mubritinib
Mubritinib (TAK-165) is a protein kinase inhibitor which was under development by Takeda for the treatment of cancer.[1][2][3] It completed phase I clinical trials but appears to have been discontinued, as no new information on the drug has surfaced since December 2008.[4]
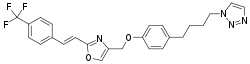 | |
| Clinical data | |
|---|---|
| Routes of administration | Oral |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C25H23F3N4O2 |
| Molar mass | 468.480 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
See also
References
- McCormick F, Fabbro D (2005). Protein Tyrosine Kinases: From Inhibitors to Useful Drugs (Cancer Drug Discovery and Development). Totowa, NJ: Humana Press. doi:10.1385/1-59259-962-1:001. ISBN 1-58829-384-X.
- Mitscher LA, Lednicer D (1977). The organic chemistry of drug synthesis. New York: Wiley. ISBN 0-470-10750-2.
- Lednicer D (2008). Strategies for Organic Drug Synthesis and Design. New York: Wiley-Interscience. ISBN 978-0-470-19039-5.
- Clinical trial number NCT00034281 for "Safety and Tolerability Study of TAK-165 in Subjects With Tumors Expressing HER2" at ClinicalTrials.gov
| CI monoclonal antibodies ("-mab") |
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Tyrosine kinase inhibitors ("-nib") |
| ||||||||
| Other |
| ||||||||
Growth factor receptor modulators | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Angiopoietin |
| ||||||||||
| CNTF |
| ||||||||||
| EGF (ErbB) |
| ||||||||||
| FGF |
| ||||||||||
| HGF (c-Met) |
| ||||||||||
| IGF |
| ||||||||||
| LNGF (p75NTR) |
| ||||||||||
| PDGF |
| ||||||||||
| RET (GFL) |
| ||||||||||
| SCF (c-Kit) | |||||||||||
| TGFβ |
| ||||||||||
| Trk |
| ||||||||||
| VEGF |
| ||||||||||
| Others |
| ||||||||||
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.