Naltrexone
Naltrexone, sold under the brand name Revia among others, is a medication primarily used to manage alcohol or opioid use disorder by reducing cravings and feelings of euphoria associated with substance use disorder.[7] It has also been found to be effective in the treatment of other addictions and may be used for them off-label.[10] An opioid-dependent person should not receive naltrexone before detoxification.[7] It is taken by mouth or by injection into a muscle.[7] Effects begin within 30 minutes.[7] A decreased desire for opioids may take a few weeks to occur.[7]
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /ˌnælˈtrɛksoʊn/ |
| Trade names | Revia, Vivitrol, Depade, others |
| Other names | EN-1639A; UM-792; ALKS-6428; N-cyclopropylmethylnoroxymorphone; N-cyclopropylmethyl-14-hydroxydihydromorphinone; 17-(cyclopropylmethyl)-4,5α-epoxy-3,14-dihydroxymorphinan-6-one |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a685041 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth, intramuscular injection, subcutaneous implant |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 5–60%[4][5] |
| Protein binding | 20%[4][1] |
| Metabolism | Liver (non-CYP450)[6] |
| Metabolites | 6β-Naltrexol, others[4] |
| Onset of action | 30 minutes[7] |
| Elimination half-life | Oral (ReVia):[1] • Naltrexone: 4 hours • 6β-Naltrexol: 13 hours Oral (Contrave):[2] • Naltrexone: 5 hours IM (Vivitrol):[3] • Naltrexone: 5–10 days • 6β-Naltrexol: 5–10 days |
| Duration of action | >72 hours[4][8][9] |
| Excretion | Urine[1] |
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.036.939 |
| Chemical and physical data | |
| Formula | C20H23NO4 |
| Molar mass | 341.407 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 169 °C (336 °F) |
SMILES
| |
InChI
| |
| (verify) | |
Side effects may include trouble sleeping, anxiety, nausea, and headaches.[7] In those still on opioids, opioid withdrawal may occur.[7] Use is not recommended in people with liver failure.[7] It is unclear if use is safe during pregnancy.[7][11] Naltrexone is an opioid antagonist and works by blocking the effects of opioids, including both opioid drugs as well as opioids naturally produced in the brain.[7]
Naltrexone was first made in 1965 and was approved for medical use in the United States in 1984.[7][12] Naltrexone, as naltrexone/bupropion (brand name Contrave), is also used to treat obesity.[13]
Medical uses
Alcohol use disorder
Naltrexone has been best studied as a treatment for alcoholism.[10] Naltrexone has been shown to decrease the quantity and frequency of ethanol consumption.[14] It does not appear to change the percentage of people drinking.[15] Its overall benefit has been described as "modest".[16]
Acamprosate may work better than naltrexone for eliminating alcohol abuse, while naltrexone may decrease the desire for alcohol to a greater extent.[17]
The Sinclair method is a method involving opiate antagonists such as naltrexone to treat alcoholism. The person takes the medication once, about an hour before consuming alcohol, to curb the urge to drink.[18][19] The opioid antagonist is thought to block the positive reinforcement effects of alcohol and may assist the person to stop or reduce their drinking.[19]
Opioid use
Long-acting injectable naltrexone is an opioid receptor antagonist, blocking the effects of heroin and other opioids, and decreases heroin use compared to a placebo.[20] Unlike methadone and buprenorphine, it is not a controlled medication.[20] It may decrease cravings for opioids after a number of weeks, and decreases the risk of overdose, at least during the time period that naltrexone is still active, though concern about risk of overdose for those stopping treatment remains.[7][21][22] It is given once per month and has better compliance and effect for opioid use than the oral formulation.[23]
A drawback of injectable naltrexone as compared to methadone and buprenorphine is the requirement for a period of opioid abstinence before starting the medication, as the injection can otherwise cause sudden and intense opioid withdrawal. This "hurdle" is responsible for worse patient uptake with long-acting injectable naltrexone as compared with buprenorphine. Among patients able to successfully initiate injectable naltrexone, long-term rates were similar [24] Another concern is that while methadone and buprenorphine patients maintain high drug tolerance in the event of return to street drug use, naltrexone allows tolerance to fade, leading to risk of overdose in people who relapse and thus higher mortality. Guidelines from the World Health Organization cite evidence of superiority in reducing mortality and retaining patients in care with opioid agonists (methadone or buprenorphine), concluding that most patients should be advised to use agonists rather than antagonists like naltrexone.[25]
A 2011 review found insufficient evidence to determine the effect of naltrexone taken by mouth in opioid dependence.[26] While some do well with this formulation, it must be taken daily, and a person whose cravings become overwhelming can obtain opioid intoxication simply by skipping a dose. Due to this issue, the usefulness of oral naltrexone in opioid use disorders is limited by the low retention in treatment. Naltrexone by mouth remains an ideal treatment for a small number of people with opioid use, usually those with a stable social situation and motivation. With additional contingency management support, naltrexone may be effective in a broader population.[27]
Others
Naltrexone is not useful for quitting smoking.[28] Naltrexone has also been under investigation for reducing behavioral addictions such as gambling or kleptomania as well as compulsive sexual behaviors in both offenders and non-offenders (e.g. compulsive porn viewing and masturbation). The results were promising. In one study, the majority of sexual offenders reported a strong reduction in sexual urges and fantasies which reverted to baseline once the medication was discontinued. Case reports have also shown cessation of gambling and other compulsive behaviors, for as long as the medication was taken.[29][30]
Available forms
Naltrexone is available and most commonly used in the form of an oral tablet (50 mg).[31] Vivitrol, a naltrexone formulation for depot injection containing 380 mg of the medication per vial, is also available.[31][32] Additionally, naltrexone subcutaneous implants that are surgically implanted are available.[33] While these are manufactured in Australia, they are not authorized for use within Australia, but only for export.[34] By 2009, naltrexone implants showed superior efficacy in the treatment of heroin dependence when compared to the oral form.[35]
When taken at much smaller doses, a regimen known as Low Dose Naltrexone (LDN), naltrexone may reduce pain and help to address neurological symptoms. Some patients report that LDN helps reduce their symptoms of ME/CFS, multiple sclerosis (MS), fibromyalgia (FMS), or autoimmune disease. Although its mechanism of action is unclear, some have speculated that it may act as an anti-inflammatory. [36] LDN is also being considered as a potential treatment for Long COVID.[37]
Contraindications
Naltrexone should not be used by persons with acute hepatitis or liver failure, or those with recent opioid use (typically 7–10 days).
Side effects
The most common side effects reported with naltrexone are gastrointestinal complaints such as diarrhea and abdominal cramping.[1] These adverse effects are analogous to the symptoms of opioid withdrawal, as the μ-opioid receptor blockade will increase gastrointestinal motility.
The side effects of naltrexone by incidence are as follows:[1]
- Greater than 10%: difficulty sleeping, anxiety, nervousness, abdominal pain/cramps, nausea and/or vomiting, low energy, joint/muscle pain, and headache.[1]
- Less than 10%: loss of appetite, diarrhea, constipation, thirstiness, increased energy, feeling down, irritability, dizziness, skin rash, delayed ejaculation, erectile dysfunction, and chills.[1]
- A variety of other adverse events have also been reported with less than 1% incidence.[1]
Opioid withdrawal
Naltrexone should not be started until several (typically 7–10) days of abstinence from opioids have been achieved. This is due to the risk of acute opioid withdrawal if naltrexone is taken, as naltrexone will displace most opioids from their receptors. The time of abstinence may be shorter than 7 days, depending on the half-life of the specific opioid taken. Some physicians use a naloxone challenge to determine whether an individual has any opioids remaining. The challenge involves giving a test dose of naloxone and monitoring for opioid withdrawal. If withdrawal occurs, naltrexone should not be started.[38]
Aversive effects
Whether naltrexone causes dysphoria, depression, anhedonia, or other aversive effects as side effects has been studied and reviewed.[39][40][41][42] In early studies in normal and opioid-abstinent individuals, acute and short-term administration of naltrexone was reported to produce a variety of aversive effects including fatigue, loss of energy, sleepiness, mild dysphoria, depression, lightheadedness, faintness, mental confusion, nausea, gastrointestinal disturbances, sweating, and occasional feelings of unreality.[41][43][44][45][46] However, these studies were small, often uncontrolled, and used subjective means of assessing side effects.[46][39] Most subsequent longer-term studies of naltrexone for indications like alcohol or opioid dependence have not reported dysphoria or depression with naltrexone in most individuals.[41][47][46] According to one source:[40]
- Naltrexone itself produces little or no psychoactive effect in normal research volunteers even at high doses, which is remarkable given that the endogenous opioid system is important in normal hedonic functioning. Because endogenous opioids are involved in the brain reward system, it would be reasonable to hypothesize that naltrexone might produce anhedonic or dysphoric effects. Although some evidence from small, early trials suggested that patients with a history of opiate dependence might be susceptible to dysphoric effects in response to naltrexone (Crowley et al. 1985; Hollister et al. 1981), reports of such effects have been inconsistent. Most large clinical studies of recovering opioid-dependent individuals have not found naltrexone to have an adverse effect on mood (Greenstein et al. 1984; Malcolm et al. 1987; Miotto et al. 2002; Shufman et al. 1994). Some studies have actually found improvements in mood during the course of treatment with naltrexone (Miotto et al. 1997; Rawlins and Randall 1976).
Based on available evidence, naltrexone seems to have minimal untoward effects in the aforementioned areas, at least with long-term therapy, .[39][40][41][42] It has been suggested that differences in findings between acute and longer-term studies of naltrexone treatment might be related to altered function in the opioid system with chronic administration of naltrexone.[41][39] For example, marked upregulation of opioid receptors and hyper-sensitivity to opioids have been observed with naltrexone in preclinical studies.[4][39][48] Another possibility is that the central opioid system may have low endogenous functionality in most individuals, becoming active only in the presence of exogenously administered opioid receptor agonists or with stimulation by endogenous opioids induced by pain or stress.[48] A third possibility is that normal individuals may experience different side effects with naltrexone than people with addictive disease such as alcohol or opioid dependence, who may have altered opioid tone or responsiveness.[39][48] It is notable in this regard that most studies of naltrexone have been in people with substance dependence.[39]
Naltrexone may also initially produce opioid withdrawal-like symptoms in a small subset of people not dependent on opioids:[49]
- The side-effect profile [of naltrexone], at least on the recommended dose of 50 mg per day, is generally benign, although 5 to 10 percent of detoxified opioid addicts experience immediate, intolerable levels of withdrawal-like effects including agitation, anxiety, insomnia, light-headedness, sweating, dysphoria, and nausea. Most patients on naltrexone experience few or no symptoms after the first 1 to 2 weeks of treatment; for a substantial minority (20 to 30 percent) protracted discomfort is experienced.
Persisting affective distress related to naltrexone may account for individuals taking the drug who drop out of treatment.[50][39][49]
Naltrexone has been reported to reduce feelings of social connection.[51][52][53][54] Studies on whether naltrexone can decrease the pleasurable effects of listening to music are conflicting.[55][56][57] Besides humans, naltrexone has been found to produce aversive effects in rodents as assessed by conditioned place aversion.[39]
Liver damage
Naltrexone has been reported to cause liver damage when given at doses higher than recommended.[58] It carries an FDA boxed warning for this rare side effect. Due to these reports, some physicians may check liver function tests prior to starting naltrexone, and periodically thereafter. Concerns for liver toxicity initially arose from a study of nonaddicted obese patients receiving 300 mg of naltrexone.[59] Subsequent studies have suggested limited or no toxicity in other patient populations and at typical recommended doses such as 50 to 100 mg/day.[58][10]
Overdose
No toxic effects have been observed with naltrexone in doses of up to 800 mg/day in clinical studies.[4][1] The largest reported overdose of naltrexone, which was 1,500 mg in a female patient and was equivalent to an entire bottle of medication (30 × 50 mg tablets), was uneventful.[60] No deaths are known to have occurred with naltrexone overdose.[61]
Pharmacology
Opioid receptor blockade
| Affinities (Ki) | Ratios | Refs | ||
|---|---|---|---|---|
| MOR | KOR | DOR | MOR:KOR:DOR | |
| 1.0 nM | 3.9 nM | 149 nM | 1:4:149 | [62] |
| 0.0825 nM | 0.509 nM | 8.02 nM | 1:6:97 | [63] |
| 0.2 nM | 0.4 nM | 10.8 nM | 1:2:54 | [64][65] |
| 0.23 nM | 0.25 nM | 38 nM | 1:1.1:165 | [66][48] |
| 0.62 nM | 1.88 nM | 12.3 nM | 1:3:20 | [67][68] |
| 0.11 nM | 0.19 nM | 60 nM | 1:1.7:545 | [69][70][71] |
Naltrexone and its active metabolite 6β-naltrexol are competitive antagonists of the opioid receptors.[72][73] Naltrexone is specifically an antagonist preferentially of the μ-opioid receptor (MOR), to a lesser extent of the κ-opioid receptor (KOR), and to a much lesser extent of the δ-opioid receptor (DOR).[72] However, naltrexone is not actually a silent antagonist of these receptors but instead acts as a weak partial agonist, with Emax values of 14 to 29% at the MOR, 16 to 39% at the KOR, and 14 to 25% at the DOR in different studies.[73][70][71] In accordance with its partial agonism, although naltrexone is described as a pure opioid receptor antagonist, it has shown some evidence of weak opioid effects in clinical and preclinical studies.[4]
By itself, naltrexone acts as an antagonist or weak partial agonist of the opioid receptors.[73] In combination with agonists of the MOR such as morphine however, naltrexone appears to become an inverse agonist of the MOR.[73] Conversely, naltrexone remains a neutral antagonist (or weak partial agonist) of the KOR and DOR.[73] In contrast to naltrexone, 6β-naltrexol is purely a neutral antagonist of the opioid receptors.[74] The MOR inverse agonism of naltrexone when it is co-present with MOR agonists may in part underlie its ability to precipitate withdrawal in opioid-dependent individuals.[74][73] This may be due to suppression of basal MOR signaling via inverse agonism.[74][73]
Occupancy of the opioid receptors in the brain by naltrexone has been studied using positron emission tomography (PET).[58][75] Naltrexone at a dose of 50 mg/day has been found to occupy approximately 90 to 95% of brain MORs and 20 to 35% of brain DORs.[58] Naltrexone at a dose of 100 mg/day has been found to achieve 87% and 92% brain occupancy of the KOR in different studies.[76][75][77] Per simulation, a lower dose of naltrexone of 25 mg/day might be expected to achieve around 60% brain occupancy of the KOR but still close to 90% occupancy of the MOR.[75] In a study of the duration of MOR blockade with naltrexone, the drug with a single 50 mg dose showed 91% blockade of brain [11C]carfentanil (a selective MOR ligand) binding at 48 hours (2 days), 80% blockade at 72 hours (3 days), 46% blockade at 120 hours (5 days), and 30% blockade at 168 hours (7 days).[8][9] The half-time of brain MOR blockade by naltrexone in this study was 72 to 108 hours (3.0 to 4.5 days).[8][9] Based on these findings, doses of naltrexone of even less than 50 mg/day would be expected to achieve virtually complete brain MOR occupancy.[8][9] Blockade of brain MORs with naltrexone is much longer-lasting than with other opioid antagonists like naloxone (half-time of ~1.7 hours intranasally) or nalmefene (half-time of ~29 hours).[8][78][79]
The half-life of occupancy of the brain MOR and duration of clinical effect of naltrexone are much longer than suggested by its plasma elimination half-life.[8][80][9][81] A single 50 mg oral dose of naltrexone has been found to block brain MORs and opioid effects for at least 48 to 72 hours.[80][9][82] The half-time of brain MOR blockade by naltrexone (72–108 hours) is much longer than the fast plasma clearance component of naltrexone and 6β-naltrexol (~4–12 hours) but was reported to correspond well to the longer terminal phase of plasma naltrexone clearance (96 hours).[8][9][39] As an alternative possibility, the prolonged brain MOR occupancy by opioid antagonists like naltrexone and nalmefene may be due to slow dissociation from MORs consequent to their very high MOR affinity (<1.0 nM).[79][83]
Naltrexone blocks the effects of MOR agonists like morphine, heroin, and hydromorphone in humans via its MOR antagonism.[4][6] Following a single 100 mg dose of naltrexone, the subjective and objective effects of heroin were blocked by 90% at 24 hours, with blockade then decreasing up to 72 hours.[4] Similarly, 20 to 200 mg naltrexone dose-dependently antagonized the effects of heroin for up to 72 hours.[4] Naltrexone also blocks the effects of KOR agonists like salvinorin A, pentazocine, and butorphanol in humans via its KOR antagonism.[84][85][86][65] In addition to opioids, naltrexone has been found to block or reduce the rewarding and other effects of other euphoriant drugs including alcohol,[48] nicotine,[87] and amphetamines.[88]
The opioid receptors are involved in neuroendocrine regulation.[4] MOR agonists produce increases in levels of prolactin and decreases in levels of luteinizing hormone (LH) and testosterone.[4] Doses of naltrexone of 25 to 150 mg/day have been found to produce significant increases in levels of β-endorphin, cortisol, and LH, equivocal changes in levels of prolactin and testosterone, and no significant changes in levels of adrenocorticotrophic hormone (ACTH) or follicle-stimulating hormone (FSH).[4] Naltrexone influences the hypothalamic–pituitary–adrenal axis (HPA axis) probably through interference with opioid receptor signaling by endorphins.[4]
Blockade of MORs is thought to be the mechanism of action of naltrexone in the management of opioid dependence—it reversibly blocks or attenuates the effects of opioids. It is also thought to be involved in the effectiveness of naltrexone in alcohol dependence by reducing the euphoric effects of alcohol. The role of KOR modulation by naltrexone in its effectiveness for alcohol dependence is unclear but this action may also be involved based on theory and animal studies.[89][90]
Other activities
In addition to the opioid receptors, naltrexone binds to and acts as an antagonist of the opioid growth factor receptor (OGFR) and toll-like receptor 4 (TLR4) and interacts with high- and low-affinity binding sites in filamin-A (FLNA).[91][92][93][94] It is said that very low doses of naltrexone (<0.001–1 mg/day) interact with FLNA, low doses (1 to 5 mg/day) produce TLR4 antagonism, and standard clinical doses (50 to 100 mg/day) exert opioid receptor and OGFR antagonism.[91][93] The interactions of naltrexone with FLNA and TLR4 are claimed to be involved in the therapeutic effects of low-dose naltrexone.[91]
Pharmacokinetics
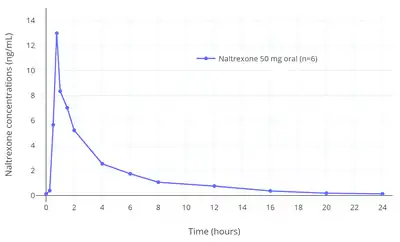
The absorption of naltrexone with oral administration is rapid and nearly complete (96%).[1] The bioavailability of naltrexone with oral administration is 5 to 60% due to extensive first-pass metabolism.[4][5] Peak concentrations of naltrexone are 19 to 44 μg/L after a single 100 mg oral dose and time to peak concentrations of naltrexone and 6β-naltrexol (metabolite) is within 1 hour.[4][5][1] Linear increases in circulating naltrexone and 6β-naltrexol concentrations occur over an oral dose range of 50 to 200 mg.[4] Naltrexone does not appear to be accumulated with repeated once-daily oral administration and there is no change in time to peak concentrations with repeated administration.[4]
The plasma protein binding of naltrexone is about 20% over a naltrexone concentration range of 0.1 to 500 μg/L.[4][1] Its apparent volume of distribution at 100 mg orally is 16.1 L/kg after a single dose and 14.2 L/kg with repeated doses.[4]
Naltrexone is metabolized in the liver mainly by dihydrodiol dehydrogenases into 6β-naltrexol (6β-hydroxynaltrexone).[4][5] Levels of 6β-naltrexol are 10- to 30-fold higher than those of naltrexone with oral administration due to extensive first-pass metabolism.[101] Conversely, 6β-naltrexol exposure is only about 2-fold higher than that of naltrexone with intramuscular injection of naltrexone in microspheres (brand name Vivitrol).[97] 6β-Naltrexol is an opioid receptor antagonist similarly to naltrexone and shows a comparable binding profile to the opioid receptors.[102] However, 6β-naltrexol is peripherally selective and crosses into the brain much less readily than does naltrexone.[102] In any case, 6β-naltrexol does still show some central activity and may contribute significantly to the central actions of oral naltrexone.[102][4] Other metabolites of naltrexone include 2-hydroxy-3-methoxy-6β-naltrexol and 2-hydroxy-3-methoxynaltrexone.[4] Following their formation, the metabolites of naltrexone are further metabolized by conjugation with glucuronic acid to form glucuronides.[4] Naltrexone is not metabolized by the cytochrome P450 system and has low potential for drug interactions.[6]
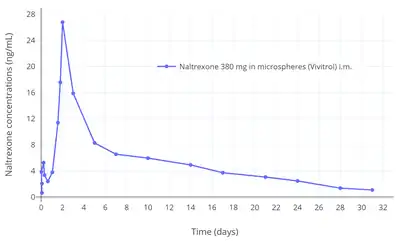
The elimination of naltrexone is biexponential and rapid over the first 24 hours followed by a third extremely slow decline after 24 hours.[4] The fast elimination half-lives of naltrexone and its metabolite 6β-naltrexol are about 4 hours and 13 hours, respectively.[1] In Contrave oral tablets, which also contain bupropion and are described as extended-release, the half-life of naltrexone is 5 hours.[2] The slow terminal-phase elimination half-life of naltrexone is approximately 96 hours.[9] As microspheres of naltrexone by intramuscular injection (Vivitrol), the elimination half-lives of naltrexone and 6β-naltrexol are both 5 to 10 days.[3] Whereas oral naltrexone is administered daily, naltrexone in microspheres by intramuscular injection is suitable for administration once every 4 weeks or once per month.[3]
Chemistry
Naltrexone, also known as N-cyclopropylmethylnoroxymorphone, is a derivative of oxymorphone (14-hydroxydihydromorphinone). It is specifically the derivative of oxymorphone in which the tertiary amine methyl substituent is replaced with methylcyclopropane.
Analogues
The closely related medication, methylnaltrexone (N-methylnaltrexone), is used to treat opioid-induced constipation, but does not treat addiction as it does not cross the blood–brain barrier. Nalmefene (6-desoxy-6-methylenenaltrexone) is similar to naltrexone and is used for the same purposes as naltrexone. Naltrexone should not be confused with naloxone (N-allylnoroxymorphone), which is used in emergency cases of opioid overdose. Other opioid antagonists related to naltrexone include 6β-naltrexol (6β-hydroxynaltrexone), samidorphan (3-carboxamido-4-hydroxynaltrexone), β-funaltrexamine (naltrexone fumarate methyl ester), nalodeine (N-allylnorcodeine), nalorphine (N-allylnormorphine), and nalbuphine (N-cyclobutylmethyl-14-hydroxydihydronormorphine).
History
Naltrexone was first synthesized in 1963 by Metossian at Endo Laboratories, a small pharmaceutical company in New York City.[105] It was characterized by Blumberg, Dayton, and Wolf in 1965 and was found to be an orally active, long-acting, and very potent opioid antagonist.[105][106][107][12] The drug showed advantages over earlier opioid antagonists such as cyclazocine, nalorphine, and naloxone, including its oral activity, a long duration of action allowing for once-daily administration, and a lack of dysphoria, and was selected for further development.[12] It was patented by Endo Laboratories in 1967 under the developmental code name EN-1639A and Endo Laboratories was acquired by DuPont in 1969.[108] Clinical trials for opioid dependence began in 1973, and a developmental collaboration of DuPont with the National Institute on Drug Abuse for this indication started the next year in 1974.[108] The drug was approved by the FDA for the oral treatment of opioid dependence in 1984, with the brand name Trexan, and for the oral treatment of alcohol dependence in 1995, when the brand name was changed by DuPont to Revia.[108][31] A depot formulation for intramuscular injection was approved by the FDA under the brand name Vivitrol for alcohol dependence in 2006 and opioid dependence in 2010.[32][31]
Society and culture
Generic names
Naltrexone is the generic name of the drug and its INN, USAN, BAN, DCF, and DCIT, while naltrexone hydrochloride is its USP and BANM.[109][110][111][112]
Brand names
Naltrexone is or has been sold under a variety of brand names, including Adepend, Antaxone, Celupan, Depade, Nalorex, Narcoral, Nemexin, Nodict, Revia, Trexan, Vivitrex, and Vivitrol.[109][110][111][112] It is also marketed in combination with bupropion (naltrexone/bupropion) as Contrave,[113] and was marketed with morphine (morphine/naltrexone) as Embeda.[112][114] A combination of naltrexone with buprenorphine (buprenorphine/naltrexone) has been developed, but has not been marketed.[115]
Controversies
The FDA authorized use of injectable naltrexone (Vivitrol) for opioid addiction using a single study[116] that was led by Evgeny Krupitsky at Bekhterev Research Psychoneurological Institute, St Petersburg State Pavlov Medical University, St Petersburg, Russia,[117] a country where opioid agonists such as methadone and buprenorphine are not available. The study was a "double-blind, placebo-controlled, randomized", 24-week trial running "from July 3, 2008, through October 5, 2009" with "250 patients with opioid dependence disorder" at "13 clinical sites in Russia" on the use of injectable naltrexone (XR-NTX) for opioid dependence. The study was funded by the Boston-based biotech Alkermes firm which produces and markets naltrexone in the United States. Critics charged that the study violated ethical guidelines, since it compared the formulation of naltrexone not to the best available, evidence-based treatment (methadone or buprenorphine), but to a placebo. Further, the trial did not follow patients who dropped out of the trial to evaluate subsequent risk of fatal overdose, a major health concern .[118] Subsequent trials in Norway and the US did compare injectable naltrexone to buprenorphine and found them to be similar in outcomes for patients willing to undergo the withdrawal symptoms required prior to naltrexone administration.[119] Nearly 30% of patients in the US trial did not complete induction.[120] In real world settings, a review of more than 40,000 patient records found that while methadone and buprenorphine reduced risk of fatal overdose, naltrexone administration showed no greater effect on overdose or subsequent emergency care than counseling alone.[22]
Despite these findings, naltrexone's manufacturer and some health authorities have promoted the medicine as superior to methadone and buprenorphine since it is not an opioid and does not induce dependence. The manufacturer has also marketed directly to law enforcement and criminal justice officials, spending millions of dollars on lobbying and providing thousands of free doses to jails and prisons.[121] The technique has been successful, with the criminal justice system in 43 states now incorporating long-acting naltrexone. Many do this through Vivitrol courts that offer only this option, leading some to characterize this as "an offer that cannot be refused."[122][123] The company's marketing techiques have led to a Congressional investigation,[124] and warning from the FDA about failure to adequately state risks of fatal overdose to patients receiving the medicine.[125]
In May 2017, United States Secretary of Health and Human Services Tom Price praised [Vivitrol] as the future of opioid addiction treatment after visiting the company's plant in Ohio.[121] His remarks set off sharp criticism with almost 700 experts in the field of substance use submitting a letter to Price cautioning him about Vivitrol's "marketing tactics" and warning him that his comments "ignore widely accepted science".[126] The experts pointed out that Vivitrol's competitors, buprenorphine and methadone, are "less expensive", "more widely used", and have been "rigorously studied". Price had claimed that buprenorphine and methadone were "simply substitute[s]" for "illicit drugs"[121] whereas according to the letter, "the substantial body of research evidence supporting these treatments is summarized in guidance from within your own agency, including the Substance Abuse and Mental Health Services Administration, the US Surgeon General, the National Institute on Drug Abuse, and the Centers for Disease Control and Prevention. Buprenorphine and methadone have been demonstrated to be highly effective in managing the core symptoms of opioid use disorder, reducing the risk of relapse and fatal overdose, and encouraging long-term recovery."[126]
Film
One Little Pill was a 2014 documentary film about use of naltrexone to treat alcohol use disorder.[127]
Research
Depersonalization
Naltrexone is sometimes used in the treatment of dissociative symptoms such as depersonalization and derealization.[128][129] Some studies suggest it might help.[130] Other small, preliminary studies have also shown benefit.[128][129] Blockade of the KOR by naltrexone and naloxone is thought to be responsible for their effectiveness in ameliorating depersonalization and derealization.[128][129] Since these drugs are less efficacious in blocking the KOR relative to the MOR, higher doses than typically used seem to be necessary.[128][129]
Low-dose
"Low-dose naltrexone" (LDN) describes the off-label use of naltrexone at low doses for diseases not related to chemical dependency or intoxication, such as multiple sclerosis.[131] Evidence for recommending such use is lacking.[132][133] This treatment has received attention on the Internet.[134] In 2022, 4 studies with a few hundred patients are experimenting using the drug for the treatment of Long COVID. [135]
Self-injury
One study suggests that self-injurious behaviors present in persons with developmental disabilities (including autism) can sometimes be remedied with naltrexone.[136] In these cases, the self-injury is believed to be done to release beta-endorphin, which binds to the same receptors as heroin and morphine.[137] If the "rush" generated by self-injury is removed, the behavior may stop.
Behavioral disorders
Some indications exist that naltrexone might be beneficial in the treatment of impulse-control disorders such as kleptomania, compulsive gambling, or trichotillomania (compulsive hair pulling), but evidence of its effectiveness for gambling is conflicting.[138][139][140] A 2008 case study reported successful use of naltrexone in suppressing and treating an internet pornography addiction.[141]
Interferon alpha
Naltrexone is effective in suppressing the cytokine-mediated adverse neuropsychiatric effects of interferon alpha therapy.[142][143]
Critical addiction studies
Some historians and sociologists have suggested that the meanings and uses attributed to anti-craving medicine, such as naltrexone, is context-dependent.[144] Studies have suggested the use of naltrexone in drug courts or healthcare rehabs is a form of "post-social control,"[145] or "post-disciplinary control,"[146] whereby control strategies for managing offenders and addicts shift from imprisonment and supervision toward more direct control over biological processes.
Sexual addiction
Small studies have shown a reduction of sexual addiction and problematic sexual behaviours from naltrexone.[147][148]
References
- "Revia (naltrexone hydrochloride tablets USP50 mgOpioid Antagonist". DailyMed. 24 April 2015. Archived from the original on 14 June 2022. Retrieved 13 June 2022.
- "Contrave Extended-Release- naltrexone hydrochloride and bupropion hydrochloride tablet, extended release". DailyMed. 4 November 2021. Archived from the original on 4 June 2020. Retrieved 13 June 2022.
- "Vivitrol- naltrexone kit". DailyMed. 10 March 2021. Archived from the original on 30 May 2022. Retrieved 13 June 2022.
- Gonzalez JP, Brogden RN (March 1988). "Naltrexone. A review of its pharmacodynamic and pharmacokinetic properties and therapeutic efficacy in the management of opioid dependence". Drugs. 35 (3): 192–213. doi:10.2165/00003495-198835030-00002. PMID 2836152.
- Lee MW, Fujioka K (August 2009). "Naltrexone for the treatment of obesity: review and update". Expert Opin Pharmacother. 10 (11): 1841–5. CiteSeerX 10.1.1.496.9477. doi:10.1517/14656560903048959. PMID 19537999. S2CID 207477935.
- Sevarino KA, Kosten TR (2009). "Naltrexone for Initiation and Maintenance of Opiate Abstinence". In Dean RL, Bilsky EJ, Negus SS (eds.). Opiate Receptors and Antagonists. Humana Press. pp. 227–245. doi:10.1007/978-1-59745-197-0_12. ISBN 978-1-58829-881-2.
- "Naltrexone Monograph for Professionals - Drugs.com". Drugs.com. American Society of Health-System Pharmacists. Archived from the original on 9 November 2017. Retrieved 9 November 2017.
- Colasanti A, Lingford-Hughes A, Nutt D (2013). "Opioids Neuroimaging". In Miller PM (ed.). Biological Research on Addiction. Comprehensive Addictive Behaviors and Disorders. Vol. 2. Elsevier. pp. 675–687. doi:10.1016/B978-0-12-398335-0.00066-2. ISBN 9780123983350.
- Lee MC, Wagner HN, Tanada S, Frost JJ, Bice AN, Dannals RF (July 1988). "Duration of occupancy of opiate receptors by naltrexone". J Nucl Med. 29 (7): 1207–11. PMID 2839637. Archived from the original on 29 October 2021. Retrieved 29 October 2021.
- Aboujaoude E, Salame WO (August 2016). "Naltrexone: A Pan-Addiction Treatment?". CNS Drugs. 30 (8): 719–33. doi:10.1007/s40263-016-0373-0. PMID 27401883. S2CID 6372144.
- Tran TH, Griffin BL, Stone RH, Vest KM, Todd TJ (July 2017). "Methadone, Buprenorphine, and Naltrexone for the Treatment of Opioid Use Disorder in Pregnant Women". Pharmacotherapy. 37 (7): 824–839. doi:10.1002/phar.1958. PMID 28543191. S2CID 13772333.
- Sadock BJ, Sadock VA, Sussman N (2012). Kaplan & Sadock's Pocket Handbook of Psychiatric Drug Treatment. Lippincott Williams & Wilkins. p. 265. ISBN 9781451154467. Archived from the original on 5 December 2017.
- "Naltrexone/bupropion for obesity". Drug and Therapeutics Bulletin. 55 (11): 126–129. November 2017. doi:10.1136/dtb.2017.11.0550. PMID 29117992. S2CID 547660.
- Rösner S, Hackl-Herrwerth A, Leucht S, Vecchi S, Srisurapanont M, Soyka M (December 2010). Srisurapanont M (ed.). "Opioid antagonists for alcohol dependence". The Cochrane Database of Systematic Reviews (12): CD001867. doi:10.1002/14651858.CD001867.pub2. PMID 21154349.
- Donoghue K, Elzerbi C, Saunders R, Whittington C, Pilling S, Drummond C (June 2015). "The efficacy of acamprosate and naltrexone in the treatment of alcohol dependence, Europe versus the rest of the world: a meta-analysis". Addiction. 110 (6): 920–30. doi:10.1111/add.12875. PMID 25664494. Archived from the original on 15 April 2019. Retrieved 15 April 2019.
- Garbutt JC (2010). "Efficacy and tolerability of naltrexone in the management of alcohol dependence". Current Pharmaceutical Design. 16 (19): 2091–7. doi:10.2174/138161210791516459. PMID 20482515.
- Maisel NC, Blodgett JC, Wilbourne PL, Humphreys K, Finney JW (February 2013). "Meta-analysis of naltrexone and acamprosate for treating alcohol use disorders: when are these medications most helpful?". Addiction. 108 (2): 275–93. doi:10.1111/j.1360-0443.2012.04054.x. PMC 3970823. PMID 23075288.
- Anderson K (28 July 2013). "Drink Your Way Sober with Naltrexone". Psychology Today. Retrieved 18 July 2016.
- Sinclair JD (2001). "Evidence about the use of naltrexone and for different ways of using it in the treatment of alcoholism". Alcohol and Alcoholism. 36 (1): 2–10. doi:10.1093/alcalc/36.1.2. PMID 11139409.
- Sharma A, Kelly SM, Mitchell SG, Gryczynski J, O'Grady KE, Schwartz RP (June 2017). "Update on Barriers to Pharmacotherapy for Opioid Use Disorders". Current Psychiatry Reports. 19 (6): 35. doi:10.1007/s11920-017-0783-9. PMC 7075636. PMID 28526967.
- Sharma B, Bruner A, Barnett G, Fishman M (July 2016). "Opioid Use Disorders". Child and Adolescent Psychiatric Clinics of North America. 25 (3): 473–87. doi:10.1016/j.chc.2016.03.002. PMC 4920977. PMID 27338968.
- Wakeman SE, Larochelle MR, Ameli O, Chaisson CE, McPheeters JT, Crown WH, et al. (February 2020). "Comparative Effectiveness of Different Treatment Pathways for Opioid Use Disorder". JAMA Network Open. 3 (2): e1920622. doi:10.1001/jamanetworkopen.2019.20622. PMID 32022884. S2CID 211035316.
- Comer SD, Sullivan MA, Yu E, Rothenberg JL, Kleber HD, Kampman K, Dackis C, O'Brien CP (February 2006). "Injectable, sustained-release naltrexone for the treatment of opioid dependence: a randomized, placebo-controlled trial". Archives of General Psychiatry. 63 (2): 210–8. doi:10.1001/archpsyc.63.2.210. PMC 4200530. PMID 16461865.
- Lee JD, Nunes EV, Novo P, Bachrach K, Bailey GL, Bhatt S, et al. (January 2018). "Comparative effectiveness of extended-release naltrexone versus buprenorphine-naloxone for opioid relapse prevention (X:BOT): a multicentre, open-label, randomised controlled trial". Lancet. 391 (10118): 309–318. doi:10.1016/S0140-6736(17)32812-X. PMC 5806119. PMID 29150198.
- Guidelines for the psychosocially assisted pharmacological treatment of opioid dependence. World Health Organization. 2009. ISBN 978-92-4-154754-3.
- Minozzi S, Amato L, Vecchi S, Davoli M, Kirchmayer U, Verster A (April 2011). Minozzi S (ed.). "Oral naltrexone maintenance treatment for opioid dependence". The Cochrane Database of Systematic Reviews (4): CD001333. doi:10.1002/14651858.CD001333.pub4. PMC 7045778. PMID 21491383.
- Johansson BA, Berglund M, Lindgren A (April 2006). "Efficacy of maintenance treatment with naltrexone for opioid dependence: a meta-analytical review". Addiction. 101 (4): 491–503. doi:10.1111/j.1360-0443.2006.01369.x. PMID 16548929.
- David SP, Lancaster T, Stead LF, Evins AE, Prochaska JJ (June 2013). "Opioid antagonists for smoking cessation". The Cochrane Database of Systematic Reviews (6): CD003086. doi:10.1002/14651858.CD003086.pub3. PMC 4038652. PMID 23744347.
- Mouaffak F, Leite C, Hamzaoui S, Benyamina A, Laqueille X, Kebir O (2017). "Naltrexone in the Treatment of Broadly Defined Behavioral Addictions: A Review and Meta-Analysis of Randomized Controlled Trials". European Addiction Research. 23 (4): 204–210. doi:10.1159/000480539. PMID 28877518.
- Ryback RS (July 2004). "Naltrexone in the treatment of adolescent sexual offenders". The Journal of Clinical Psychiatry. 65 (7): 982–986. doi:10.4088/jcp.v65n0715. PMID 15291688.
- Milhorn HT (17 October 2017). Substance Use Disorders: A Guide for the Primary Care Provider. Springer International Publishing. pp. 88–. ISBN 978-3-319-63040-3. Archived from the original on 27 April 2021. Retrieved 4 December 2017.
- "Alcoholism Once A Month Injectable Drug, Vivitrol, Approved By FDA Archived 2009-01-05 at the Wayback Machine," Medical News Today, 16 April 2006.
- Therapeutic Goods Administration. "Australian Register of Therapeutic Goods Medicines" (Online database of approved medicines). Archived from the original on 14 May 2009. Retrieved 22 March 2009.
- Therapeutic Goods Administration. "Australian Register of Therapeutic Goods Medicines" (Online database of approved medicines, specific entry for "O'Neil Long Acting Naltrexone Implant"). Retrieved 27 April 2017.
- Hulse GK, Morris N, Arnold-Reed D, Tait RJ (October 2009). "Improving clinical outcomes in treating heroin dependence: randomized, controlled trial of oral or implant naltrexone". Archives of General Psychiatry. 66 (10): 1108–15. doi:10.1001/archgenpsychiatry.2009.130. PMID 19805701.
- "Low dose naltrexone - MEpedia".
- O'Kelly B, Vidal L, McHugh T, Woo J, Avramovic G, Lambert JS (October 2022). "Safety and efficacy of low dose naltrexone in a long covid cohort; an interventional pre-post study". Brain, Behavior, & Immunity - Health. 24: 100485. doi:10.1016/j.bbih.2022.100485. PMC 9250701. PMID 35814187.
- Galanter M, Kleber HD, eds. (2008). The American Psychiatric Publishing Textbook of Substance Abuse Treatment. American Psychiatric. ISBN 978-1-58562-276-4.
- Miotto K, McCann M, Basch J, Rawson R, Ling W (2002). "Naltrexone and dysphoria: fact or myth?". Am J Addict. 11 (2): 151–60. doi:10.1080/10550490290087929. PMID 12028745.
- Strain EC, Stitzer ML (2006). The Treatment of Opioid Dependence. JHU Press. pp. 296–. ISBN 978-0-8018-8219-7. Archived from the original on 2 June 2021. Retrieved 2 June 2021.
- Nathan PJ, O'Neill BV, Napolitano A, Bullmore ET (October 2011). "Neuropsychiatric adverse effects of centrally acting antiobesity drugs". CNS Neurosci Ther. 17 (5): 490–505. doi:10.1111/j.1755-5949.2010.00172.x. PMC 6493804. PMID 21951371.
- Krupitsky E, Zvartau E, Blokhina E, Verbitskaya E, Wahlgren V, Tsoy-Podosenin M, Bushara N, Burakov A, Masalov D, Romanova T, Tyurina A, Palatkin V, Yaroslavtseva T, Pecoraro A, Woody G (September 2016). "Anhedonia, depression, anxiety, and craving in opiate dependent patients stabilized on oral naltrexone or an extended release naltrexone implant". Am J Drug Alcohol Abuse. 42 (5): 614–620. doi:10.1080/00952990.2016.1197231. PMC 5156574. PMID 27436632.
- Mendelson JH, Ellingboe J, Keuhnle JC, Mello NK (October 1978). "Effects of naltrexone on mood and neuroendocrine function in normal adult males". Psychoneuroendocrinology. 3 (3–4): 231–6. doi:10.1016/0306-4530(78)90013-6. PMID 219434. S2CID 7712730.
- Hollister LE, Johnson K, Boukhabza D, Gillespie HK (August 1981). "Aversive effects of naltrexone in subjects not dependent on opiates". Drug Alcohol Depend. 8 (1): 37–41. doi:10.1016/0376-8716(81)90084-3. PMID 7297411.
- Crowley TJ, Wagner JE, Zerbe G, Macdonald M (September 1985). "Naltrexone-induced dysphoria in former opioid addicts". Am J Psychiatry. 142 (9): 1081–4. doi:10.1176/ajp.142.9.1081. PMID 2992300.
- Malcolm R, O'Neil PM, Von JM, Dickerson PC (June 1987). "Naltrexone and dysphoria: a double-blind placebo controlled trial". Biol Psychiatry. 22 (6): 710–6. doi:10.1016/0006-3223(87)90202-2. PMID 3593812. S2CID 39628172.
- Pettinati HM, O'Brien CP, Rabinowitz AR, Wortman SP, Oslin DW, Kampman KM, Dackis CA (December 2006). "The status of naltrexone in the treatment of alcohol dependence: specific effects on heavy drinking". J Clin Psychopharmacol. 26 (6): 610–25. doi:10.1097/01.jcp.0000245566.52401.20. PMID 17110818. S2CID 35171287.
- Unterwald EM (September 2008). "Naltrexone in the treatment of alcohol dependence". J Addict Med. 2 (3): 121–7. doi:10.1097/ADM.0b013e318182b20f. PMID 21768981. S2CID 23603792.
- Rounsaville BJ (1995). "Can psychotherapy rescue naltrexone treatment of opioid addiction?" (PDF). NIDA Res Monogr. 150: 37–52. PMID 8742771. Archived (PDF) from the original on 19 January 2022. Retrieved 31 October 2021.
- Carroll KM, Nich C, Frankforter TL, Yip SW, Kiluk BD, DeVito EE, Sofuoglu M (November 2018). "Accounting for the uncounted: Physical and affective distress in individuals dropping out of oral naltrexone treatment for opioid use disorder". Drug Alcohol Depend. 192: 264–270. doi:10.1016/j.drugalcdep.2018.08.019. PMC 6203294. PMID 30300800.
- Inagaki TK, Hazlett LI, Andreescu C (April 2020). "Opioids and social bonding: Effect of naltrexone on feelings of social connection and ventral striatum activity to close others". J Exp Psychol Gen. 149 (4): 732–745. doi:10.1037/xge0000674. PMC 7021584. PMID 31414860.
- Inagaki TK, Hazlett LI, Andreescu C (May 2019). "Naltrexone alters responses to social and physical warmth: implications for social bonding". Soc Cogn Affect Neurosci. 14 (5): 471–479. doi:10.1093/scan/nsz026. PMC 6545530. PMID 30976797.
- Inagaki TK, Ray LA, Irwin MR, Way BM, Eisenberger NI (May 2016). "Opioids and social bonding: naltrexone reduces feelings of social connection". Soc Cogn Affect Neurosci. 11 (5): 728–35. doi:10.1093/scan/nsw006. PMC 4847702. PMID 26796966.
- Meier IM, Bos PA, Hamilton K, Stein DJ, van Honk J, Malcolm-Smith S (December 2016). "Naltrexone increases negatively-valenced facial responses to happy faces in female participants". Psychoneuroendocrinology. 74: 65–68. doi:10.1016/j.psyneuen.2016.08.022. PMID 27588701. S2CID 40097592.
- O'Brien CP, Gastfriend DR, Forman RF, Schweizer E, Pettinati HM (2011). "Long-term opioid blockade and hedonic response: preliminary data from two open-label extension studies with extended-release naltrexone". Am J Addict. 20 (2): 106–12. doi:10.1111/j.1521-0391.2010.00107.x. PMC 3895092. PMID 21314752.
- Mallik A, Chanda ML, Levitin DJ (February 2017). "Anhedonia to music and mu-opioids: Evidence from the administration of naltrexone". Sci Rep. 7: 41952. Bibcode:2017NatSR...741952M. doi:10.1038/srep41952. PMC 5296903. PMID 28176798.
- Laeng B, Garvija L, Løseth G, Eikemo M, Ernst G, Leknes S (April 2021). "'Defrosting' music chills with naltrexone: The role of endogenous opioids for the intensity of musical pleasure". Conscious Cogn. 90: 103105. doi:10.1016/j.concog.2021.103105. PMID 33711654. S2CID 232163311.
- Ray LA, Chin PF, Miotto K (March 2010). "Naltrexone for the treatment of alcoholism: clinical findings, mechanisms of action, and pharmacogenetics". CNS Neurol Disord Drug Targets. 9 (1): 13–22. doi:10.2174/187152710790966704. PMID 20201811.
- Pfohl DN, Allen JI, Atkinson RL, Knopman DS, Malcolm RJ, Mitchell JE, Morley JE (1986). "Naltrexone hydrochloride (Trexan): a review of serum transaminase elevations at high dosage". NIDA Research Monograph. 67: 66–72. PMID 3092099. Archived from the original on 21 January 2017. Retrieved 23 January 2017.
- Reece AS (September 2010). "Clinical safety of 1500 mg oral naltrexone overdose". BMJ Case Rep. 2010: bcr0420102871. doi:10.1136/bcr.04.2010.2871. PMC 3028212. PMID 22778191.
- Pettinati HM, Dundon WD, Casares López MJ (2013). "Naltrexone and Opioid Antagonists for Alcohol Dependence". Interventions for Addiction. Elsevier. pp. 375–384. doi:10.1016/B978-0-12-398338-1.00039-7. ISBN 978-0-12-398338-1.
- Raynor K, Kong H, Chen Y, Yasuda K, Yu L, Bell GI, Reisine T (February 1994). "Pharmacological characterization of the cloned κ-, δ-, and μ-opioid receptors". Molecular Pharmacology. 45 (2): 330–4. CiteSeerX 10.1.1.1076.4629. PMID 8114680. INIST:3935705. Archived from the original on 22 June 2018. Retrieved 22 June 2018.
- Codd EE, Shank RP, Schupsky JJ, Raffa RB (September 1995). "Serotonin and norepinephrine uptake inhibiting activity of centrally acting analgesics: structural determinants and role in antinociception". The Journal of Pharmacology and Experimental Therapeutics. 274 (3): 1263–70. PMID 7562497. Archived from the original on 2 February 2017. Retrieved 23 January 2017.
- Toll L, Berzetei-Gurske IP, Polgar WE, Brandt SR, Adapa ID, Rodriguez L, Schwartz RW, Haggart D, O'Brien A, White A, Kennedy JM, Craymer K, Farrington L, Auh JS (March 1998). "Standard binding and functional assays related to medications development division testing for potential cocaine and opiate narcotic treatment medications". NIDA Res Monogr. 178: 440–66. CiteSeerX 10.1.1.475.3403. PMID 9686407.
- Clark SD, Abi-Dargham A (October 2019). "The Role of Dynorphin and the Kappa Opioid Receptor in the Symptomatology of Schizophrenia: A Review of the Evidence". Biol Psychiatry. 86 (7): 502–511. doi:10.1016/j.biopsych.2019.05.012. PMID 31376930. S2CID 162168648.
- Peng X, Knapp BI, Bidlack JM, Neumeyer JL (May 2007). "Pharmacological properties of bivalent ligands containing butorphan linked to nalbuphine, naltrexone, and naloxone at mu, delta, and kappa opioid receptors". J Med Chem. 50 (9): 2254–8. doi:10.1021/jm061327z. PMC 3357624. PMID 17407276.
- Zheng MQ, Nabulsi N, Kim SJ, Tomasi G, Lin SF, Mitch C, Quimby S, Barth V, Rash K, Masters J, Navarro A, Seest E, Morris ED, Carson RE, Huang Y (March 2013). "Synthesis and evaluation of 11C-LY2795050 as a κ-opioid receptor antagonist radiotracer for PET imaging". J Nucl Med. 54 (3): 455–63. doi:10.2967/jnumed.112.109512. PMC 3775344. PMID 23353688.
- Cumming P, Marton J, Lilius TO, Olberg DE, Rominger A (November 2019). "A Survey of Molecular Imaging of Opioid Receptors". Molecules. 24 (22): 4190. doi:10.3390/molecules24224190. PMC 6891617. PMID 31752279.
- Wentland MP, Lu Q, Lou R, Bu Y, Knapp BI, Bidlack JM (April 2005). "Synthesis and opioid receptor binding properties of a highly potent 4-hydroxy analogue of naltrexone". Bioorg Med Chem Lett. 15 (8): 2107–10. doi:10.1016/j.bmcl.2005.02.032. PMID 15808478.
- Wentland MP, Lou R, Lu Q, Bu Y, Denhardt C, Jin J, Ganorkar R, VanAlstine MA, Guo C, Cohen DJ, Bidlack JM (April 2009). "Syntheses of novel high affinity ligands for opioid receptors". Bioorg Med Chem Lett. 19 (8): 2289–94. doi:10.1016/j.bmcl.2009.02.078. PMC 2791460. PMID 19282177.
- Linda P. Dwoskin, ed. (29 January 2014). Emerging Targets and Therapeutics in the Treatment of Psychostimulant Abuse. Academic Press. pp. 398–. ISBN 978-0-12-420177-4. OCLC 1235841274. Archived from the original on 27 April 2021. Retrieved 30 October 2021.
- Niciu MJ, Arias AJ (October 2013). "Targeted opioid receptor antagonists in the treatment of alcohol use disorders". CNS Drugs. 27 (10): 777–87. doi:10.1007/s40263-013-0096-4. PMC 4600601. PMID 23881605.
- Wang D, Sun X, Sadee W (May 2007). "Different effects of opioid antagonists on mu-, delta-, and kappa-opioid receptors with and without agonist pretreatment". J Pharmacol Exp Ther. 321 (2): 544–52. doi:10.1124/jpet.106.118810. PMID 17267582. S2CID 28500012.
- Sadée W, Wang D, Bilsky EJ (February 2005). "Basal opioid receptor activity, neutral antagonists, and therapeutic opportunities". Life Sci. 76 (13): 1427–37. doi:10.1016/j.lfs.2004.10.024. PMID 15680308.
- de Laat B, Nabulsi N, Huang Y, O'Malley SS, Froehlich JC, Morris ED, Krishnan-Sarin S (June 2020). "Occupancy of the kappa opioid receptor by naltrexone predicts reduction in drinking and craving". Mol Psychiatry. 26 (9): 5053–5060. doi:10.1038/s41380-020-0811-8. PMID 32541931. S2CID 219692020.
- Placzek MS (August 2021). "Imaging Kappa Opioid Receptors in the Living Brain with Positron Emission Tomography". Handb Exp Pharmacol. Handbook of Experimental Pharmacology. 271: 547–577. doi:10.1007/164_2021_498. ISBN 978-3-030-89073-5. PMID 34363128. S2CID 236947969.
- Vijay A, Morris E, Goldberg A, Petrulli J, Liu H, Huang Y, Krishnan-Sarin S (1 April 2017). "Naltrexone occupancy at kappa opioid receptors investigated in alcoholics by PET occupancy at kappa opioid receptors investigated in alcoholics by PET". Journal of Nuclear Medicine. 58 (Supplement 1): 1297. Archived from the original on 29 October 2021. Retrieved 29 October 2021.
- Waarde AV, Absalom AR, Visser AK, Dierckx RA (30 September 2020). "Positron Emission Tomography (PET) Imaging of Opioid Receptors". In Dierckx RA, Otte A, de Vries EF, van Waarde A, Lammertsma AA (eds.). PET and SPECT of Neurobiological Systems. Springer International Publishing. pp. 749–807. doi:10.1007/978-3-030-53176-8_21. ISBN 978-3-030-53175-1. S2CID 241535315.
- Soyka M, Rösner S (November 2010). "Nalmefene for treatment of alcohol dependence". Expert Opin Investig Drugs. 19 (11): 1451–9. doi:10.1517/13543784.2010.522990. PMID 20868291. S2CID 9227860.
- Mannelli P, Peindl KS, Wu LT (June 2011). "Pharmacological enhancement of naltrexone treatment for opioid dependence: a review". Subst Abuse Rehabil. 2011 (2): 113–123. doi:10.2147/SAR.S15853. PMC 3128868. PMID 21731898.
- Schuh KJ, Walsh SL, Stitzer ML (July 1999). "Onset, magnitude and duration of opioid blockade produced by buprenorphine and naltrexone in humans". Psychopharmacology (Berl). 145 (2): 162–74. doi:10.1007/s002130051045. PMID 10463317. S2CID 5930936.
- Narcotic Antagonists: Naltrexone Pharmacochemistry and Sustained-release Preparations. Department of Health and Human Services, Public Health Service, Alcohol, Drug Abuse, and Mental Health Administration, National Institute on Drug Abuse, Division of Research. 1981. pp. 148–. Archived from the original on 2 June 2021. Retrieved 2 June 2021.
- Ingman K, Hagelberg N, Aalto S, Någren K, Juhakoski A, Karhuvaara S, Kallio A, Oikonen V, Hietala J, Scheinin H (December 2005). "Prolonged central mu-opioid receptor occupancy after single and repeated nalmefene dosing". Neuropsychopharmacology. 30 (12): 2245–53. doi:10.1038/sj.npp.1300790. PMID 15956985. S2CID 2453226.
- Maqueda AE, Valle M, Addy PH, Antonijoan RM, Puntes M, Coimbra J, Ballester MR, Garrido M, González M, Claramunt J, Barker S, Lomnicka I, Waguespack M, Johnson MW, Griffiths RR, Riba J (July 2016). "Naltrexone but Not Ketanserin Antagonizes the Subjective, Cardiovascular, and Neuroendocrine Effects of Salvinorin-A in Humans". Int J Neuropsychopharmacol. 19 (7): pyw016. doi:10.1093/ijnp/pyw016. PMC 4966277. PMID 26874330.
- Walsh SL, Chausmer AE, Strain EC, Bigelow GE (January 2008). "Evaluation of the mu and kappa opioid actions of butorphanol in humans through differential naltrexone blockade". Psychopharmacology (Berl). 196 (1): 143–55. doi:10.1007/s00213-007-0948-z. PMC 2766188. PMID 17909753.
- Preston KL, Bigelow GE (February 1993). "Differential naltrexone antagonism of hydromorphone and pentazocine effects in human volunteers". J Pharmacol Exp Ther. 264 (2): 813–23. PMID 7679737.
- Modesto-Lowe V, Van Kirk J (August 2002). "Clinical uses of naltrexone: a review of the evidence". Exp Clin Psychopharmacol. 10 (3): 213–27. doi:10.1037/1064-1297.10.3.213. PMID 12233982.
- Lam L, Anand S, Li X, Tse ML, Zhao JX, Chan EW (April 2019). "Efficacy and safety of naltrexone for amfetamine and methamfetamine use disorder: a systematic review of randomized controlled trials". Clin Toxicol (Phila). 57 (4): 225–233. doi:10.1080/15563650.2018.1529317. PMID 30451013. S2CID 53951406.
- Soyka M, Friede M, Schnitker J (March 2016). "Comparing Nalmefene and Naltrexone in Alcohol Dependence: Are there any Differences? Results from an Indirect Meta-Analysis". Pharmacopsychiatry. 49 (2): 66–75. doi:10.1055/s-0035-1565184. PMID 26845589.
- Koob GF, Volkow ND (August 2016). "Neurobiology of addiction: a neurocircuitry analysis". The Lancet. Psychiatry. 3 (8): 760–73. doi:10.1016/S2215-0366(16)00104-8. PMC 6135092. PMID 27475769.
- Toljan K, Vrooman B (September 2018). "Low-Dose Naltrexone (LDN)-Review of Therapeutic Utilization". Med Sci (Basel). 6 (4): 82. doi:10.3390/medsci6040082. PMC 6313374. PMID 30248938.
- Bachtell R, Hutchinson MR, Wang X, Rice KC, Maier SF, Watkins LR (2015). "Targeting the Toll of Drug Abuse: The Translational Potential of Toll-Like Receptor 4". CNS Neurol Disord Drug Targets. 14 (6): 692–9. doi:10.2174/1871527314666150529132503. PMC 5548122. PMID 26022268.
- Lee B, Elston DM (June 2019). "The uses of naltrexone in dermatologic conditions". J Am Acad Dermatol. 80 (6): 1746–1752. doi:10.1016/j.jaad.2018.12.031. PMID 30582992. S2CID 58595160.
- Zagon IS, Verderame MF, McLaughlin PJ (February 2002). "The biology of the opioid growth factor receptor (OGFr)". Brain Res Brain Res Rev. 38 (3): 351–76. doi:10.1016/s0165-0173(01)00160-6. PMID 11890982. S2CID 37812525.
- Dean RL (January 2005). "The preclinical development of Medisorb Naltrexone, a once a month long acting injection, for the treatment of alcohol dependence". Front Biosci. 10 (1–3): 643–55. doi:10.2741/1559. PMID 15569605.
- Goonoo N, Bhaw-Luximon A, Ujoodha R, Jhugroo A, Hulse GK, Jhurry D (June 2014). "Naltrexone: a review of existing sustained drug delivery systems and emerging nano-based systems". J Control Release. 183: 154–66. doi:10.1016/j.jconrel.2014.03.046. PMID 24704710.
- Dunbar JL, Turncliff RZ, Dong Q, Silverman BL, Ehrich EW, Lasseter KC (March 2006). "Single- and multiple-dose pharmacokinetics of long-acting injectable naltrexone". Alcohol Clin Exp Res. 30 (3): 480–90. doi:10.1111/j.1530-0277.2006.00052.x. PMID 16499489.
- Swainston Harrison T, Plosker GL, Keam SJ (2006). "Extended-release intramuscular naltrexone". Drugs. 66 (13): 1741–51. doi:10.2165/00003495-200666130-00006. PMID 16978037. S2CID 21309382.
- Mannelli P, Peindl K, Masand PS, Patkar AA (October 2007). "Long-acting injectable naltrexone for the treatment of alcohol dependence". Expert Rev Neurother. 7 (10): 1265–77. doi:10.1586/14737175.7.10.1265. PMID 17939765. S2CID 36453900.
- "Vivitrol (naltrexone for extended-release injectable suspension)". ALK-VIV. 28 November 2018. Archived from the original on 31 October 2021. Retrieved 31 October 2021.
- Davis MP, Glare PA, Hardy J (28 May 2009). Opioids in Cancer Pain. Oxford University Press. pp. 41–. ISBN 978-0-19-923664-0. Archived from the original on 31 October 2021. Retrieved 31 October 2021.
- Hipkin RW, Dolle RE (2010). Opioid Receptor Antagonists for Gastrointestinal Dysfunction. Annual Reports in Medicinal Chemistry. Vol. 45. pp. 142–155. doi:10.1016/S0065-7743(10)45009-5. ISBN 978-0-12-380902-5.
- Ray LA, Barr CS, Blendy JA, Oslin D, Goldman D, Anton RF (March 2012). "The role of the Asn40Asp polymorphism of the mu opioid receptor gene (OPRM1) on alcoholism etiology and treatment: a critical review". Alcoholism: Clinical and Experimental Research. 36 (3): 385–94. doi:10.1111/j.1530-0277.2011.01633.x. PMC 3249007. PMID 21895723.
- Garbutt JC, Greenblatt AM, West SL, Morgan LC, Kampov-Polevoy A, Jordan HS, Bobashev GV (August 2014). "Clinical and biological moderators of response to naltrexone in alcohol dependence: a systematic review of the evidence". Addiction. 109 (8): 1274–84. doi:10.1111/add.12557. PMID 24661324.
- National Research Council (U.S.). Committee on Problems of Drug Dependence (1974). Report of the Thirty-sixth Annual Scientific Meeting: Committee on Problems of Drug Dependence, Mexico City, March 10-14, 1974. National Academies. pp. 265–. ISBN 9780309022446. NAP:13963. Archived from the original on 27 April 2021. Retrieved 4 December 2017.
- Padwa HM, Cunningham J (2010). Addiction: A Reference Encyclopedia. ABC-CLIO. pp. 207–. ISBN 978-1-59884-229-6.
- Bennett G (14 January 2004). Treating Drug Abusers. Routledge. pp. 112–. ISBN 978-1-134-93173-6. Archived from the original on 27 April 2021. Retrieved 4 December 2017.
- Wouk J (1 March 2009). Google Ldn !. Lulu.com. pp. 78–88. ISBN 978-0-578-00439-6. Archived from the original on 28 April 2021. Retrieved 4 December 2017.
- Elks J (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 851–. ISBN 978-1-4757-2085-3. Archived from the original on 6 August 2020. Retrieved 4 December 2017.
- Index Nominum 2000: International Drug Directory. Taylor & Francis. 2000. pp. 715–. ISBN 978-3-88763-075-1. Archived from the original on 6 August 2020. Retrieved 4 December 2017.
- Morton IK, Hall JM (6 December 2012). Concise Dictionary of Pharmacological Agents: Properties and Synonyms. Springer Science & Business Media. pp. 189–. ISBN 978-94-011-4439-1.
- "Naltrexone". Archived from the original on 4 December 2017. Retrieved 4 December 2017.
- "Bupropion and naltrexone Uses, Side Effects & Warnings". Drugs.com. 8 June 2020. Archived from the original on 26 October 2020. Retrieved 16 September 2020.
- "Morphine and naltrexone Uses, Side Effects & Warnings". Drugs.com. 14 October 2019. Archived from the original on 25 October 2020. Retrieved 16 September 2020.
- McCann DJ (April 2008). "Potential of buprenorphine/naltrexone in treating polydrug addiction and co-occurring psychiatric disorders". Clinical Pharmacology and Therapeutics. 83 (4): 627–30. doi:10.1038/sj.clpt.6100503. PMID 18212797. S2CID 21165673.
- Armstrong W (7 May 2013). "A Shot in the Dark: Can Vivitrol Help Us Control Our Addictions?". Pacific Standard. Archived from the original on 13 September 2017.
- Krupitsky E, Nunes EV, Ling W, Illeperuma A, Gastfriend DR, Silverman BL (April 2011). "Injectable extended-release naltrexone for opioid dependence: a double-blind, placebo-controlled, multicentre randomised trial". Lancet. 377 (9776): 1506–1513. doi:10.1016/s0140-6736(11)60358-9. PMID 21529928. S2CID 16690413.
- Wolfe D, Carrieri MP, Dasgupta N, Bruce D, Wodak A (2011). "Injectable extended-release naltrexone for opioid dependence – Authors' reply". The Lancet. 378 (9792): 666. doi:10.1016/S0140-6736(11)61333-0. S2CID 205963967.
- Tanum L, Solli KK, Latif ZE, Benth JŠ, Opheim A, Sharma-Haase K, et al. (December 2017). "Effectiveness of Injectable Extended-Release Naltrexone vs Daily Buprenorphine-Naloxone for Opioid Dependence: A Randomized Clinical Noninferiority Trial". JAMA Psychiatry. 74 (12): 1197–1205. doi:10.1001/jamapsychiatry.2017.3206. PMC 6583381. PMID 29049469.
- Lee JD, Nunes EV, Novo P, Bachrach K, Bailey GL, Bhatt S, et al. (January 2018). "Comparative effectiveness of extended-release naltrexone versus buprenorphine-naloxone for opioid relapse prevention (X:BOT): a multicentre, open-label, randomised controlled trial". Lancet. 391 (10118): 309–318. doi:10.1016/S0140-6736(17)32812-X. PMC 5806119. PMID 29150198.
- Goodnough A, Zernike K (11 June 2017). "Seizing on Opioid Crisis, a Drug Maker Lobbies Hard for Its Product". The New York Times. Archived from the original on 11 June 2017. Retrieved 11 June 2017.
Advertising for Vivitrol on a subway car in Brooklyn last month. Marketing for the drug has shifted into high gear.
- MacGillis A. "The Last Shot". ProPublica. Archived from the original on 28 December 2021. Retrieved 29 December 2021.
- Wolfe D, Saucier R (February 2021). "Biotechnologies and the future of opioid addiction treatments". The International Journal on Drug Policy. 88: 103041. doi:10.1016/j.drugpo.2020.103041. PMID 33246267. S2CID 227191111.
- Harper J (7 November 2017). "Kamala Harris Investigating Addiction Drug Manufacturer Alkermes". WFYI Public Media. Archived from the original on 14 June 2022. Retrieved 29 December 2021.
- Office of the FDA Commissioner (24 March 2020). "FDA issues warning letter for not including the most serious risks in advertisement for medication-assisted treatment drug". Food and Drug Administration. Archived from the original on 29 December 2021. Retrieved 29 December 2021.
- "Letter to Tom Price". May 2017. Archived from the original on 25 June 2017. Retrieved 11 June 2017.
- One Little Pill (2014) at IMDb
- Simeon D, Abugel J (10 October 2008). Feeling Unreal: Depersonalization Disorder and the Loss of the Self. Oxford University Press. pp. 166–. ISBN 978-0-19-976635-2. Archived from the original on 15 February 2017. Retrieved 4 October 2016.
- Lanius UF, Paulsen SL, Corrigan FM (13 May 2014). Neurobiology and Treatment of Traumatic Dissociation: Towards an Embodied Self. Springer Publishing Company. pp. 489–. ISBN 978-0-8261-0632-2. Archived from the original on 15 February 2017. Retrieved 4 October 2016.
- Sierra M (January 2008). "Depersonalization disorder: pharmacological approaches". Expert Review of Neurotherapeutics. 8 (1): 19–26. doi:10.1586/14737175.8.1.19. PMID 18088198. S2CID 22180718.
- Novella S (5 May 2010). "Low Dose Naltrexone – Bogus or Cutting Edge Science?". Science-Based Medicine. Archived from the original on 8 July 2011. Retrieved 5 July 2011.
- "Low-Dose Naltrexone". National MS Society. Archived from the original on 13 May 2014. Retrieved 12 May 2014.
- Novella S (5 May 2010). "Low Dose Naltrexone – Bogus or Cutting Edge Science?". Archived from the original on 8 July 2011. Retrieved 5 July 2011.
- Bowling AC. "Low-dose naltrexone (LDN) The "411" on LDN". National Multiple Sclerosis Society. Archived from the original on 15 May 2009. Retrieved 6 July 2011. Alt URL Archived 6 January 2021 at the Wayback Machine
- Steenhuysen, Julie (18 October 2022). "Addiction drug shows promise lifting long COVID brain fog, fatigue". Reuters.
- Smith SG, Gupta KK, Smith SH (1995). "Effects of naltrexone on self-injury, stereotypy, and social behavior of adults with developmental disabilities". Journal of Developmental and Physical Disabilities. 7 (2): 137–46. doi:10.1007/BF02684958. S2CID 144215400.
- Manley C (20 March 1998). "Self-injuries may have biochemical base: study". The Reporter. Archived from the original on 5 January 2009.
- Grant JE, Kim SW, Odlaug BL (April 2009). "A double-blind, placebo-controlled study of the opiate antagonist, naltrexone, in the treatment of kleptomania". Biological Psychiatry. 65 (7): 600–6. doi:10.1016/j.biopsych.2008.11.022. PMID 19217077. S2CID 22992128. Lay summary – Science Daily (3 April 2009).
{{cite journal}}: Cite uses deprecated parameter|lay-url=(help) - Clinical trial number NCT00326807 for "A Randomized, Double-Blind, Placebo-Controlled Trial of Naltrexone in the Treatment of Concurrent Alcohol Dependence and Pathological Gambling" at ClinicalTrials.gov
- Kim SW, Grant JE, Adson DE, Shin YC (June 2001). "Double-blind naltrexone and placebo comparison study in the treatment of pathological gambling". Biological Psychiatry. 49 (11): 914–21. doi:10.1016/S0006-3223(01)01079-4. PMID 11377409. S2CID 22134798.
- Bostwick JM, Bucci JA (February 2008). "Internet sex addiction treated with naltrexone". Mayo Clinic Proceedings. 83 (2): 226–30. doi:10.4065/83.2.226. PMID 18241634.
- Vignau J, Karila L, Costisella O, Canva V (2005). "[Hepatitis C, interferon a and depression: main physiopathologic hypothesis]" [Hepatitis C, interferon a and depression: main physiopathologic hypothesis]. L'Encéphale (in French). 31 (3): 349–57. doi:10.1016/s0013-7006(05)82400-5. PMID 16142050. INIST:16920336.
- Małyszczak K, Inglot M, Pawłowski T, Czarnecki M, Rymer W, Kiejna A (2006). "[Neuropsychiatric symptoms related to interferon alpha]" [Neuropsychiatric symptoms related to interferon alpha]. Psychiatria Polska (in Polish). 40 (4): 787–97. PMID 17068950. Archived from the original on 2 February 2017.
- Campbell ND (28 March 2013). "Why Can't They Stop?" A Highly Public Misunderstanding of Science. pp. 238–262. doi:10.1215/9780822395874-010. ISBN 978-0-8223-5350-8. Archived from the original on 14 June 2022. Retrieved 20 December 2020.
- Vrecko S (1 June 2009). "Therapeutic Justice in Drug Courts: Crime, Punishment and Societies of Control". Science as Culture. 18 (2): 217–232. doi:10.1080/09505430902885623. S2CID 144197523.
- Aleksanyan J (9 May 2020). "Governing beyond the closet: Remaking stigma, identity, and sexual behavior in a post-disciplinary rehab". Ethnography: 1466138120923702. doi:10.1177/1466138120923702. S2CID 218936388.
- Malandain L, Blanc JV, Ferreri F, Thibaut F (May 2020). "Pharmacotherapy of Sexual Addiction". Current Psychiatry Reports. 22 (6): 30. doi:10.1007/s11920-020-01153-4. PMID 32377953. S2CID 218527367.
- Herron, Abigail J., Brennan, Tim K. eds. ASAM Essentials of Addiction Medicine, The. 3rd Edition. Two Commerce Square, 2001 Market Street, Philadelphia, PA 19103 USA:Lippincott Williams & Wilkins; 2020.