Alcaftadine
 | |
| Names | |
|---|---|
| Trade names | Lastacaft |
IUPAC name
| |
| Clinical data | |
| Drug class | H1 histamine receptor blocker[1] |
| Main uses | Allergic conjunctivitis[1] |
| Side effects | Eye irritation, redness, and itchiness[1] |
| WHO AWaRe | UnlinkedWikibase error: ⧼unlinkedwikibase-error-statements-entity-not-set⧽ |
| Pregnancy category |
|
| Routes of use | Eye drop |
| Onset of action | Within 3 min[3] |
| Duration of action | Up to 16 hrs[3] |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a611022 |
| Legal | |
| License data |
|
| Legal status |
|
| Pharmacokinetics | |
| Elimination half-life | ~2 hrs |
| Chemical and physical data | |
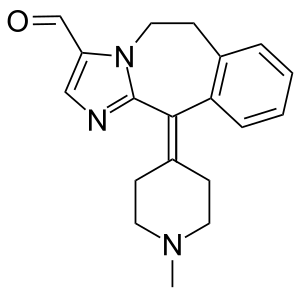
| Formula | C19H21N3O |
| Molar mass | 307.397 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
Alcaftadine, sold under the brand name Lastacaft, is a medication used to treat allergic conjunctivitis.[1] It may be used in those over the age of 1 year.[3] It is used as an eye drop.[1] Benefits occur within 3 minutes and may last for up to 16 hours.[3]
Common side effects include eye irritation, redness, and itchiness.[1] Safety in pregnancy is unclear.[4] It is a H1 histamine receptor blocker.[1]
Alcaftadine was approved for medical use in the United States in 2010.[1] In the United States 3 ml costs about 240 USD as of 2022.[5]
Medical use
When alcaftadine was tested against placebo and olopatadine, only alcaftadine 0.25% was shown to have a clinically significant reduction in conjunctival redness scores 7 and 15 minutes after administration. When clinical groups where compared to placebo, treatment with all three of the alcaftadine groups (0.05, 0.1 and 0.25%) showed a reduction in secondary endpoints of (lid swelling, conjunctival redness, ocular itching/tearing) as compared to placebo.[6]
Side effects
In studies comparing the effectiveness of olopatadine to alcaftadine, there was not a dose response increase of adverse effects as alcaftadine doses increases for 0.05% to 0.1% to 0.25%. The most common seen side effect of alcaftadine administration was irritation or a stinging sensation at the administration site.[6]
Pharmacology
Alcaftadine is an antagonist at three of the histamine receptors (1, 2 and 4). The main indication for Alcaftadine is for prevention of allergic conjunctivitis. By blocking these three receptors, Alcaftadine has been shown to significantly reduce the effects of allergens. This effect on histamine receptors seems to show lower rates of itching, eosinophil recruitment and redness after exposure to an allergen.[6] The effect of alcaftadine seemed to reduce the amount of eosinophil cells significantly as compared to olopatadine 0.1%. When animal models were used to test alcaftadine, alcaftadine 0.25% seemed to show superior results for decreasing E-cadherin expression as compared to placebo. The reduction of E-cadherin means decreased junctions which would lead to progression of allergic conjunctivitis.[7]
Pharmacokinetics
Alcaftadine is typically administrated as an eye drop which keeps its effects regional as compared to systemic effects. The main metabolite of alcaftadine is a carboxylic acid metabolite that has minimal systemic effects. The maximum concentration of its main active metabolite is 0.06 ng/ml at Tmax of 15 minutes. The minimal absorption of alcaftadine, leads to minimal systemic accumulation. The half life of the major metabolite of alcaftadine is two hours after ocular administration. Except for the metabolite, the rest of alcaftadine is excreted unchanged. Since alcaftadine is administrated at a small concentration and at a local site (the eye), it seems to have minimal systemic effects.[8]
Society and culture
Sales
Sales for alcaftadine from the company Allergan began in July 2010. Since the beginning of sales, prescriptions have increased until at least March 2012. From the time period of July 2010 until March 2012, cumulative sales have reached 139,000 prescriptions. Out of these 139,000 prescriptions there have been 104,000 unique patients. In March 2012, Alcaftadine exceeded sales of Elestat and this trend may continue as prescribers see the benefit of Alcaftadine and become more comfortable prescribing it.[9]
References
- 1 2 3 4 5 6 7 8 "DailyMed - LASTACAFT- alcaftadine solution/ drops". dailymed.nlm.nih.gov. Archived from the original on 27 September 2020. Retrieved 13 January 2022.
- ↑ "Alcaftadine ophthalmic (Lastacaft) Use During Pregnancy". Drugs.com. 11 October 2019. Archived from the original on 10 June 2020. Retrieved 10 June 2020.
- 1 2 3 4 "Alcaftadine Monograph for Professionals". Drugs.com. Archived from the original on 4 March 2021. Retrieved 13 January 2022.
- ↑ "Alcaftadine ophthalmic (Lastacaft) Use During Pregnancy". Drugs.com. Archived from the original on 10 June 2020. Retrieved 13 January 2022.
- ↑ "Alcaftadine Prices, Coupons & Savings Tips - GoodRx". GoodRx. Retrieved 13 January 2022.
- 1 2 3 Greiner JV, Edwards-Swanson K, Ingerman A (January 2011). "Evaluation of alcaftadine 0.25% ophthalmic solution in acute allergic conjunctivitis at 15 minutes and 16 hours after instillation versus placebo and olopatadine 0.1%". Clinical Ophthalmology. 5: 87–93. doi:10.2147/OPTH.S15379. PMC 3037035. PMID 21339800.
- ↑ Ono SJ, Lane K (February 2011). "Comparison of effects of alcaftadine and olopatadine on conjunctival epithelium and eosinophil recruitment in a murine model of allergic conjunctivitis". Drug Design, Development and Therapy. 5: 77–84. doi:10.2147/DDDT.S15788. PMC 3038998. PMID 21340041.
- ↑ "Alcaftadine" (PDF). Office of Clinical Pharmacology Review. U.S. Food and Drug Administration. 17 November 2009. Archived (PDF) from the original on 25 April 2019. Retrieved 9 August 2020.
- ↑ "Lastacaft (alcaftadine ophthalmic solution 0.25%)" (PDF). Drug Use Review. U.S. Food and Drug Administration. 21 June 2012. Archived from the original (PDF) on 15 February 2017.
External links
| External sites: |
|
|---|---|
| Identifiers: |