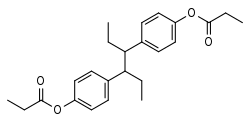
Hexestrol dipropionate
 | |
| Clinical data | |
|---|---|
| Other names | Hexestrol 4,4'-dipropionate |
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C24H30O4 |
| Molar mass | 382.500 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
Hexestrol dipropionate (brand name Hexanoestrol, Retalon Oleosum), or hexestrol dipropanoate, is a synthetic, nonsteroidal estrogen of the stilbestrol group related to diethylstilbestrol.[1] It is an ester of hexestrol,[1] and has been known since at least 1931.[2] The drug has been used in the past to inhibit lactation in women.[3][4]
| Estrogen | Form | Major brand name(s) | EPD (14 days) | Duration | |
|---|---|---|---|---|---|
| Diethylstilbestrol (DES) | Oil solution | Metestrol | 20 mg | 1 mg ≈ 2–3 days; 3 mg ≈ 3 days | |
| Diethylstilbestrol dipropionate | Oil solution | Cyren B | 12.5–15 mg | 2.5 mg ≈ 5 days | |
| Aqueous suspension | ? | 5 mg | ? mg = 21–28 days | ||
| Dimestrol (DES dimethyl ether) | Oil solution | Depot-Cyren, Depot-Oestromon, Retalon Retard | 20–40 mg | ? | |
| Fosfestrol (DES diphosphate)a | Aqueous solution | Honvan | ? | <1 day | |
| Dienestrol diacetate | Aqueous suspension | Farmacyrol-Kristallsuspension | 50 mg | ? | |
| Hexestrol dipropionate | Oil solution | Hormoestrol, Retalon Oleosum | 25 mg | ? | |
| Hexestrol diphosphatea | Aqueous solution | Cytostesin, Pharmestrin, Retalon Aquosum | ? | Very short | |
| Note: All by intramuscular injection unless otherwise noted. Footnotes: a = By intravenous injection. Sources: See template. | |||||
See also
References
- 1 2 Elks J, Ganellin CR, Elks J, eds. (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. p. 163. doi:10.1007/978-1-4757-2085-3. ISBN 978-1-4757-2085-3. OCLC 898564124.
- ↑ Report. U.S. Government Printing Office. 1931. p. 104. LCCN sn85063598.
- ↑ Prescott F, Basden M (September 1944). "Inhibition of Lactation by Hexoestrol Dipropionate". British Medical Journal. 2 (4369): 428–30. doi:10.1136/bmj.2.4369.428. PMC 2286347. PMID 20785672.
- ↑ Thomas CC, ed. (1945). Journal of Clinical Endocrinology. p. 194. ISSN 0368-1610. LCCN 45029631. OCLC 1607514.
This article is issued from Offline. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.