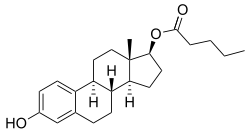
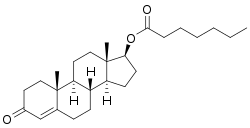
Estradiol valerate/testosterone enanthate
Estradiol valerate/testosterone enanthate (EV/TE), sold under the brand names Primodian Depot and Ditate among others, is an injectable combination medication of estradiol valerate (EV), an estrogen, and testosterone enanthate (TE), an androgen/anabolic steroid, which is used in menopausal hormone therapy for women.[1][2] The medication is also used to suppress lactation in postpartum women (brand name Deladumone).[3][4][5][6]
 | |
 | |
| Combination of | |
|---|---|
| Estradiol valerate | Estrogen |
| Testosterone enanthate | Androgen; Anabolic steroid |
| Clinical data | |
| Trade names | Deladumone, Despamen, Ditate, Ditate-DS, Gravignost, Primodian Depot, Valertest, others |
| Other names | EV/TE; TEEV |
| Routes of administration | Intramuscular injection |
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| UNII | |
Ditate was provided in the form of multi-use 10 mL vials containing 4 mg/mL EV and 90 mg/mL TE in an oil solution and was administered by intramuscular injection at regular intervals.[7] Conversely, another preparation, Ditate-DS, was provided in the form of single-use 2 mL vials containing 8 mg/mL EV and 180 mg/mL TE in an oil solution, and was administered as a single intramuscular injection.[7] Another product, Primodian Depot, was provided in the form of 1 mL ampoules containing 4 mg/mL EV (3.0 mg/mL free estradiol) and 90.3 mg/mL TE (65 mg/mL free testosterone) in an oil solution, and was administered by intramuscular injection once every 4 to 6 weeks.[8][9][10][11] The elimination half-life of EV in oil by intramuscular injection is approximately 4 or 5 days.[12] Similarly, the elimination half-life of TE in oil by intramuscular injection is approximately 4 or 5 days.[13] EV/TE reportedly has a duration of about 21 days.[14]
Deladumone OB was a double-dosage formulation of Deladumone, which was intended to provide the same dosage with a smaller injection volume.[15]
Primodian Depot was introduced for medical use by 1955.[16] An oral tablet product with the similar brand name of Primodian, containing ethinylestradiol and methyltestosterone, was marketed around the same time, and should not be confused with the injectable Primodian Depot.[8] EV/TE was discontinued in the United States by 2011.[17] EV/TE has been discontinued in most other countries as well, but formulations of EV/TE continue to be marketed in a few countries, including Japan, Mexico, Peru, and Taiwan.[18][19][20]
| Route | Medication | Major brand names | Form | Dosage |
|---|---|---|---|---|
| Oral | Testosterone undecanoate | Andriol, Jatenzo | Capsule | 40–80 mg 1x/1–2 days |
| Methyltestosterone | Metandren, Estratest | Tablet | 0.5–10 mg/day | |
| Fluoxymesterone | Halotestin | Tablet | 1–2.5 mg 1x/1–2 days | |
| Normethandronea | Ginecoside | Tablet | 5 mg/day | |
| Tibolone | Livial | Tablet | 1.25–2.5 mg/day | |
| Prasterone (DHEA)b | – | Tablet | 10–100 mg/day | |
| Sublingual | Methyltestosterone | Metandren | Tablet | 0.25 mg/day |
| Transdermal | Testosterone | Intrinsa | Patch | 150–300 μg/day |
| AndroGel | Gel, cream | 1–10 mg/day | ||
| Vaginal | Prasterone (DHEA) | Intrarosa | Insert | 6.5 mg/day |
| Injection | Testosterone propionatea | Testoviron | Oil solution | 25 mg 1x/1–2 weeks |
| Testosterone enanthate | Delatestryl, Primodian Depot | Oil solution | 25–100 mg 1x/4–6 weeks | |
| Testosterone cypionate | Depo-Testosterone, Depo-Testadiol | Oil solution | 25–100 mg 1x/4–6 weeks | |
| Testosterone isobutyratea | Femandren M, Folivirin | Aqueous suspension | 25–50 mg 1x/4–6 weeks | |
| Mixed testosterone esters | Climacterona | Oil solution | 150 mg 1x/4–8 weeks | |
| Omnadren, Sustanon | Oil solution | 50–100 mg 1x/4–6 weeks | ||
| Nandrolone decanoate | Deca-Durabolin | Oil solution | 25–50 mg 1x/6–12 weeks | |
| Prasterone enanthatea | Gynodian Depot | Oil solution | 200 mg 1x/4–6 weeks | |
| Implant | Testosterone | Testopel | Pellet | 50–100 mg 1x/3–6 months |
| Notes: Premenopausal women produce about 230 ± 70 μg testosterone per day (6.4 ± 2.0 mg testosterone per 4 weeks), with a range of 130 to 330 μg per day (3.6–9.2 mg per 4 weeks). Footnotes: a = Mostly discontinued or unavailable. b = Over-the-counter. Sources: See template. | ||||
References
- M. Notelovitz; P.A. van Keep (6 December 2012). The Climacteric in Perspective: Proceedings of the Fourth International Congress on the Menopause, held at Lake Buena Vista, Florida, October 28–November 2, 1984. Springer Science & Business Media. pp. 397–. ISBN 978-94-009-4145-8.
- Kahr, H.; Müller, H. A. (1956). "Das Klimakterium". Konservative Therapie der Frauenkrankheiten. pp. 85–102. doi:10.1007/978-3-7091-5694-0_2. ISBN 978-3-7091-5696-4.
- McNeilly, A. S. (2011). "Physiology of lactation". Journal of Biosocial Science. 9 (S4): 5–21. doi:10.1017/S0021932000023804. ISSN 0021-9320. S2CID 24614184.
- Vorherr H (July 1972). "Suppression of postpartum lactation". Postgrad Med. 52 (1): 145–52. doi:10.1080/00325481.1972.11713186. PMID 5037562.
- Helmuth Vorherr (2 December 2012). The Breast: Morphology, Physiology, and Lactation. Elsevier Science. pp. 200–203. ISBN 978-0-323-15726-1.
- Copeland Jr, W. E. (1986). Lactation Suppressants. Drug Therapy in Obstetrics and Gynecology, 241.
- de Haen, Paul (1972). "Drugs Released for Clinical Use". The Journal of Clinical Pharmacology and New Drugs. 12 (1): 58–60. doi:10.1002/j.1552-4604.1972.tb00038.x. ISSN 0021-9754.
- Hans Hermann Julius Hager; Walther Kern; Paul Heinz List; Hermann Josef Roth (29 July 2013). Hagers Handbuch der Pharmazeutischen Praxis: Für Apotheker, Arzneimittelhersteller, Ärzte und Medizinalbeamte: Wirkstoffgruppen II Chemikalien und Drogen (A-AL). Springer-Verlag. pp. 142, 156, 185. ISBN 978-3-662-25655-8.
- A. Saure (11 November 2013). Die Wechseljahre der Frau: Hormone — Präparate — Therapien. Springer-Verlag. pp. 157–. ISBN 978-3-0348-6676-7.
- Georg Arends; Heinrich Zörnig; Hermann Hager; Georg Frerichs, Walther Kern (14 December 2013). Hagers Handbuch der pharmazeutischen Praxis: Für Apotheker, Arzneimittelhersteller, Drogisten, Ärzte u. Medizinalbeamte. Springer-Verlag. pp. 1164–. ISBN 978-3-662-36329-4.
- E. Buchborn; H. Jahrmärker; H.J. Karl; G.A. Martini, W. Müller, G. Riecker, H. Schwiegk, W. Siegenthaler, W. Stich (2 July 2013). Therapie innerer Krankheiten. Springer-Verlag. pp. 405–. ISBN 978-3-662-10489-7.
{{cite book}}: CS1 maint: multiple names: authors list (link) - Düsterberg B, Nishino Y (December 1982). "Pharmacokinetic and pharmacological features of oestradiol valerate". Maturitas. 4 (4): 315–24. doi:10.1016/0378-5122(82)90064-0. PMID 7169965.
- Luetjens, Craig Marc; Wistuba, Joachim; Weinbauer, Gerhard; Nieschlag, Eberhard (2007). "The Leydig Cell as a Target for Male Contraception". The Leydig Cell in Health and Disease. Contemporary Endocrinology. pp. 415–442. doi:10.1007/978-1-59745-453-7_29. ISBN 978-1-58829-754-9.
- Ufer, Joachim (1 January 1978). Hormontherapie in der Frauenheilkunde: Grundlagen und Praxis [Hormone Therapy in Gynecology: Principles and Practice] (in German) (5 ed.). de Gruyter. p. 276. ISBN 978-3110066647. OCLC 924728827.
- Physicians Desk Reference to Pharmaceutical Specialties and Biologicals. Medical Economics. 1971. p. 1262.
Deladumone OB contains the same hormonal agents as Deladumone (Testosterone Enanthate and Estradiol Valerate Injection) but at twice the potency, in order to provide a high dosage concentration in a low volume.
- Geburtshilfe und Frauenheilkunde: Ergebnisse der Forschung für die Praxis. Georg Thieme Verlag. 1955. pp. 1061–1062.
- Food and Drug Administration (2011). Approved Drug Products with Therapeutic Equivalence Evaluations - FDA Orange Book 31st Edition (2011): FDA Orange Book 31st Edition (2011). DrugPatentWatch.com. pp. 586–. ISBN 978-1-934899-81-6.
- "Estradiol". Drugs.com.
- Sweetman, Sean C., ed. (2009). "Sex hormones and their modulators". Martindale: The Complete Drug Reference (36th ed.). London: Pharmaceutical Press. pp. 2100, 2124–2125. ISBN 978-0-85369-840-1.
- "IBM Watson Health Products: Please Login". www.micromedexsolutions.com.