Δ-10-Tetrahydrocannabinol
 | |
 | |
| Identifiers | |
|---|---|
IUPAC name
| |
| CAS Number |
|
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| Chemical and physical data | |
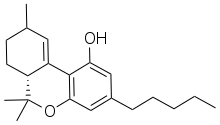
| Formula | C21H30O2 |
| Molar mass | 314.469 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
Δ-10-Tetrahydrocannabinol (Delta-10-THC, Δ10-THC, alternatively numbered as Δ2-THC) is a positional isomer of tetrahydrocannabinol, discovered in the 1980s. Two enantiomers have been reported in the literature, with the 9-methyl group in either the (R) or (S) conformation; of these, the (R) enantiomer appears to be the more active isomer as well as the double bond in the 10th position instead of the 9th maintaining about 30 to 40 percent the potency of delta-9-THC.[1][2][3] Δ10-THC has rarely been reported as a trace component of natural cannabis, though it is thought to be a degradation product similar to cannabinol rather than being produced by the plant directly. However, it is found more commonly as an impurity in synthetic delta-8-THC produced from cannabidiol[4][5][6] and can also be synthesized directly from delta-9-THC.[7]
See also
References
- ↑ Srebnik M, Lander N, Breuer A, Mechoulam R (1984). "Base-catalysed double-bond isomerizations of cannabinoids: structural and stereochemical aspects". Journal of the Chemical Society, Perkin Transactions 1: 2881–6. doi:10.1039/P19840002881.
- ↑ Järbe TU, Hiltunen AJ, Mechoulam R, Srebnik M, Breuer A (November 1988). "Separation of the discriminative stimulus effects of stereoisomers of delta 2- and delta 3-tetrahydrocannabinols in pigeons". European Journal of Pharmacology. 156 (3): 361–6. doi:10.1016/0014-2999(88)90281-6. PMID 2850933.
- ↑ WHO Expert Committee on Drug Dependence Critical Review. Isomers of THC (PDF) (Report). World Health Organization. 2018.
- ↑ Hanuš LO, Meyer SM, Muñoz E, Taglialatela-Scafati O, Appendino G (November 2016). "Phytocannabinoids: a unified critical inventory". Natural Product Reports. 33 (12): 1357–1392. doi:10.1039/c6np00074f. PMID 27722705.
- ↑ Chan-Hosokawa A, Nguyen L, Lattanzio N, Adams WR (March 2021). "Emergence of Delta-8 Tetrahydrocannabinol (THC) in DUID Investigation Casework: Method Development, Validation and Application". Journal of Analytical Toxicology. 46 (1): 1–9. doi:10.1093/jat/bkab029. PMID 33754645.
- ↑ Erickson BE. Delta-8-THC craze concerns chemists. Chemical & Engineering News, 30 August 2021
- ↑ WO 2020248059, Adair C, Geiling B, Haghdoost Manjili M., "Methods for preparing cannabinoids by base-promoted double-bond migration.", published 2020-12-17, assigned to Canopy Growth Corporation