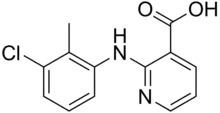
Clonixin
 | |
| Names | |
|---|---|
| Preferred IUPAC name
2-(3-Chloro-2-methylanilino)pyridine-3-carboxylic acid | |
| Other names
Clonixic acid; CBA 93626[1] | |
| Identifiers | |
CAS Number |
|
3D model (JSmol) |
|
| ChEBI |
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.037.921 |
| KEGG | |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula |
C13H11ClN2O2 |
| Molar mass | 262.69 g·mol−1 |
| Pharmacology | |
| per os | |
| Pharmacokinetics: | |
| Glucuronidation via UGT2B7 | |
| Legal status |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Clonixin is a nonsteroidal anti-inflammatory drug (NSAID). It also has analgesic, antipyretic, and platelet-inhibitory actions. It is used primarily in the treatment of chronic arthritic conditions and certain soft tissue disorders associated with pain and inflammation.
Synthesis
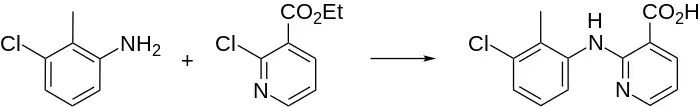
Clonixin synthesis: M. H. Sherlock, N. Sperber, BE 679271; eidem, U.S. Patent 3,337,570 (1966, 1967 both to Schering).
Clonixeril
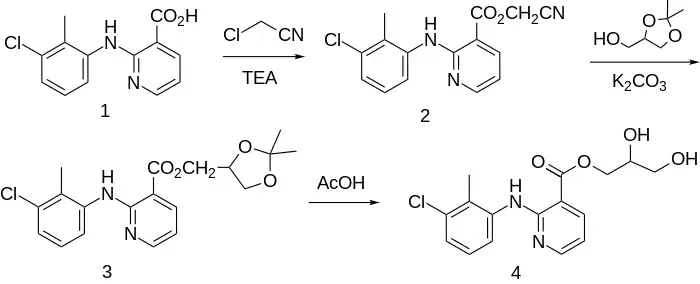
The glyceryl ester of clonixin, clonixeril, is also a NSAID. It was prepared by a somewhat roundabout method.
Clonixin was reacted with chloroacetonitrile and triethylamine to give 2. Heating with potassium carbonate and glycerol actonide displaced the activating group to produce ester 3, which was deblocked in acetic acid to produce clonixeril (4).
See also
- Flunixin
- Clonixeril
References
- ↑ Finch, Jay S.; Dekornfeld, Thomas J. (1971). "Clonixin: A Clinical Evaluation of a New Oral Analgesic". The Journal of Clinical Pharmacology and New Drugs. 11 (5): 371–377. doi:10.1177/009127007101100508 (inactive 31 October 2021). PMID 4935715. Retrieved 2015-05-21.
{{cite journal}}: CS1 maint: DOI inactive as of October 2021 (link) - ↑ M. H. Sherlock, ZA 6802185 (1968); Chem. Abstr., 70: 96640c (1969).
- ↑ CH 534129 (1973); Chem. Abstr., 79: 18582g (1973).
This article is issued from Offline. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.