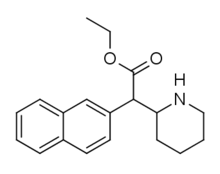
HDEP-28
HDEP-28 or ethylnaphthidate is a piperidine based stimulant drug, closely related to ethylphenidate, but with the benzene ring replaced by naphthalene. It is even more closely related to HDMP-28, which acts as a potent serotonin–norepinephrine–dopamine reuptake inhibitor with several times the potency of methylphenidate and a short duration of action.[1] It has been sold as a designer drug since around 2015.[2]
 | |
| Legal status | |
|---|---|
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| Chemical and physical data | |
| Formula | C19H23NO2 |
| Molar mass | 297.398 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Legality
HDEP-28 was banned in the UK as a Temporary Class Drug from June 2015 following its unapproved sale as a designer drug, alongside 4-Methylmethylphenidate.[3][4][5]
See also
References
- Lile JA, Wang Z, Woolverton WL, France JE, Gregg TC, Davies HM, Nader MA (October 2003). "The reinforcing efficacy of psychostimulants in rhesus monkeys: the role of pharmacokinetics and pharmacodynamics". The Journal of Pharmacology and Experimental Therapeutics. 307 (1): 356–66. doi:10.1124/jpet.103.049825. PMID 12954808. S2CID 5654856.
- Luethi D, Kaeser PJ, Brandt SD, Krähenbühl S, Hoener MC, Liechti ME. Pharmacological profile of methylphenidate-based designer drugs. Neuropharmacology. 2018 May 15;134(Pt A):133-140. doi:10.1016/j.neuropharm.2017.08.020 PMID 28823611
- Methylphenidate-based NPS: A review of the evidence of use and harm. Advisory Council on the Misuse of Drugs, 31 March 2015
- "Letter to Mike Penning on methylphenidate-based novel psychoactive substances". Advisory Council on the Misuse of Drugs. 25 June 2015. Retrieved 25 June 2015.
- "Ministerial response to the Advisory Council on the Misuse of Drugs about 2 new methylphenidate-based substances". Home Office. 25 June 2015. Retrieved 25 June 2015.
| D1-like |
| ||||||
|---|---|---|---|---|---|---|---|
| D2-like |
| ||||||
| |||||||
| 5-HT1 |
| ||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 5-HT2 |
| ||||||||||||||||||||||||||||||||||||||
| 5-HT3–7 |
| ||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.