MDAT
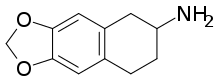
6,7-Methylenedioxy-2-aminotetralin (MDAT) is a drug developed in the 1990s by a team at Purdue University led by David E. Nichols.[1] It appears to act as a serotonin releasing agent based on rodent drug discrimination assays comparing it to MDMA, in which it fully substitutes for, and additionally lacks any kind of serotonergic neurotoxicity.[1] Hence, MDAT is considered likely to be a non-neurotoxic, putative entactogen in humans.
 | |
| Clinical data | |
|---|---|
| Routes of administration | Oral |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
IUPAC name
| |
| CAS Number |
|
| PubChem CID | |
| ChemSpider | |
| UNII |
|
| Chemical and physical data | |
| Formula | C11H13NO2 |
| Molar mass | 191.230 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
See also
- 2-Aminotetralin
References
- Nichols DE, Brewster WK, Johnson MP, Oberlender R, Riggs RM (February 1990). "Nonneurotoxic tetralin and indan analogues of 3,4-(methylenedioxy)amphetamine (MDA)". Journal of Medicinal Chemistry. 33 (2): 703–10. doi:10.1021/jm00164a037. PMID 1967651.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.