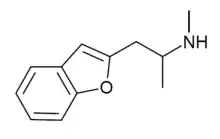
2-MAPB
2-MAPB is a recreational designer drug with empathogenic effects. As with other related substituted benzofuran derivatives such as 6-APB and 5-MAPB, 2-MAPB is a monoamine releaser with some selectivity for serotonin release, generally similar in pharmacological profile to MDMA but with greater activity as a directly acting agonist of 5-HT2 receptor subtypes and somewhat greater toxicity.[1] 2-MAPB has been isolated from post-mortem toxicology screens in several drug-related fatal adverse reactions but generally only as a component of combinations of drugs, making it difficult to determine how much it contributed to the deaths.[2][3] It is illegal in Japan.[4]
 | |
| Identifiers | |
|---|---|
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| Chemical and physical data | |
| Formula | C12H15NO |
| Molar mass | 189.258 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
References
- Fuwa T, Suzuki J, Tanaka T, Inomata A, Honda Y, Kodama T (2016). "Novel psychoactive benzofurans strongly increase extracellular serotonin level in mouse corpus striatum". The Journal of Toxicological Sciences. 41 (3): 329–337. doi:10.2131/jts.41.329. PMID 27193726.
- Staeheli SN, Boxler MI, Oestreich A, Marti M, Gascho D, Bolliger SA, et al. (October 2017). "Postmortem distribution and redistribution of MDAI and 2-MAPB in blood and alternative matrices" (PDF). Forensic Science International. 279: 83–87. doi:10.1016/j.forsciint.2017.08.007. PMID 28850871.
- Theofel N, Budach D, Vejmelka E, Scholtis S, Tsokos M (June 2021). "Toxicological investigations in a death involving 2-MAPB". Forensic Science, Medicine, and Pathology. 17 (2): 317–321. doi:10.1007/s12024-021-00366-0. ISSN 1547-769X. PMID 33877515. S2CID 233309570.
- "指定薬物名称・構造式一覧(平成27年9月16日現在)" (PDF) (in Japanese). 厚生労働省. 16 September 2015. Retrieved 23 August 2016.
Empathogens/entactogens | |
|---|---|
| Phenylalkyl- amines (other than cathinones) |
|
| Cyclized phenyl- alkylamines | |
| Cathinones |
|
| Tryptamines | |
| Chemical classes | |
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.