Alfuzosin
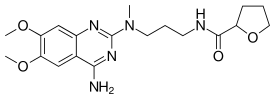 | |
| Names | |
|---|---|
| Pronunciation | /ælˈfjuːzoʊsɪn/ al-FEW-zoh-sin |
| Trade names | Uroxatral, others |
IUPAC name
| |
| Clinical data | |
| Drug class | α1 adrenergic receptor blocker[1] |
| Main uses | Benign prostatic hyperplasia (BPH)[1] |
| Side effects | Lightheadedness, tiredness, headache[1] |
| WHO AWaRe | UnlinkedWikibase error: ⧼unlinkedwikibase-error-statements-entity-not-set⧽ |
| Pregnancy category |
|
| Routes of use | By mouth (tablets) |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a64002 |
| Legal | |
| Legal status | |
| Pharmacokinetics | |
| Bioavailability | 49% |
| Protein binding | 82–90% |
| Metabolism | Liver (CYP3A4-mediated) |
| Elimination half-life | 10 hours |
| Excretion | Feces (69%) and Urine (24%) |
| Chemical and physical data | |
| Formula | C19H27N5O4 |
| Molar mass | 389.456 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
Alfuzosin, sold under the brand name Uroxatral among others, is a medication used to treat benign prostatic hyperplasia (BPH).[1] It may be less effective than other medications in its class.[2] It may be used together with a 5α-reductase inhibitors.[1] It is taken by mouth once per day.[1]
Common side effects include lightheadedness, tiredness, and headache.[1] Other side effects include low blood pressure and QT prolongation.[1] Use is not recommended in those with significant liver problems.[1] It is a α1 adrenergic receptor blocker and works by relaxing the muscles in the prostate and bladder neck.[1]
Alfuzosin was patented in 1978 and approved for medical use in 1988.[3] It was approved for medical use in the United States in 2003.[1] In the United States it costs about 18 USD for 3 months.[4] In 2017, it was the 266th most commonly prescribed medication in the United States, with more than one million prescriptions.[5][6]
Medical uses
It is used to treat benign prostatic hyperplasia (BPH).[1] It may be less effective than other medications in its class.[2] It may be used together with a 5α-reductase inhibitors.[1] It is taken by mouth once per day.[1]
Use is not recommended for high blood pressure.[1]
Side effects
The most common side effects are dizziness (due to postural hypotension), upper respiratory tract infection, headache, fatigue, and abdominal disturbances. Side effects include stomach pain, heartburn, and congested nose.[7] Adverse effects of alfuzosin are similar to that of tamsulosin with the exception of retrograde ejaculation.[8]
Contraindications
Alfuzosin should be used with caution in people with severe chronic kidney disease, and should not be prescribed to patients with a known history of QT prolongation or those who are taking medications known to prolong the QT interval.
Chemistry
Alfuzosin contains a stereocenter and is therefore chiral. There are two enantiomeric forms, (R)-alfuzosin and (S)-alfuzosin. The drug is used as a racemate, (RS)-alfuzosin, a 1: 1 mixture of the (R)- and (S)-forms.[9]
| Enantiomers of alfuzosin | |
|---|---|
-Alfuzosin_Structural_Formula_V2.svg.png.webp) CAS number: 123739-69-5 |
-Alfuzosin_Structural_Formula_V2.svg.png.webp) CAS number.: 123739-70-8 |
It is provided as the hydrochloride salt.
Society and culture
Cost
In 2017, it was the 266th most commonly prescribed medication in the United States, with more than one million prescriptions.[5][6]
.svg.png.webp) Alfuzosin costs (US)
Alfuzosin costs (US).svg.png.webp) Alfuzosin prescriptions (US)
Alfuzosin prescriptions (US)
Brand names
It is marketed under the brand name Uroxatral, Xat, Xatral, Prostetrol and Alfural.
References
- 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 "Alfuzosin Monograph for Professionals". Drugs.com. Archived from the original on 19 January 2021. Retrieved 14 July 2021.
- 1 2 Lepor H (August 2016). "Alpha-blockers for the Treatment of Benign Prostatic Hyperplasia". The Urologic Clinics of North America. 43 (3): 311–23. doi:10.1016/j.ucl.2016.04.009. PMC 2213889. PMID 27476124.
- ↑ Fischer J, Ganellin CR (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 455. ISBN 9783527607495. Archived from the original on 2020-10-05. Retrieved 2020-09-19.
- ↑ "Alfuzosin Prices, Coupons & Patient Assistance Programs". Drugs.com. Archived from the original on 5 July 2017. Retrieved 14 July 2021.
- 1 2 "The Top 300 of 2020". ClinCalc. Archived from the original on 18 March 2020. Retrieved 11 April 2020.
- 1 2 "Alfuzosin Hydrochloride - Drug Usage Statistics". ClinCalc. Archived from the original on 12 April 2020. Retrieved 11 April 2020.
- ↑ "Alfuzosin". MedlinePlus. United States National Library of Medicine. April 15, 2016. Archived from the original on August 24, 2019. Retrieved January 16, 2018.
- ↑ Liu C, Zeng G, Kang R, Wu W, Li J, Chen K, Wan SP (2015). "Efficacy and Safety of Alfuzosin as Medical Expulsive Therapy for Ureteral Stones: A Systematic Review and Meta-Analysis". PLOS ONE. 10 (8): e0134589. Bibcode:2015PLoSO..1034589L. doi:10.1371/journal.pone.0134589. PMC 4526635. PMID 26244843.
 This article incorporates text available under the CC BY 4.0 license.
This article incorporates text available under the CC BY 4.0 license. - ↑ Rote Liste Service GmbH (Hrsg.): Rote Liste 2017 - Arzneimittelverzeichnis für Deutschland (einschließlich EU-Zulassungen und bestimmter Medizinprodukte). Rote Liste Service GmbH, Frankfurt/Main, 2017, Aufl. 57, S. 159, ISBN 978-3-946057-10-9.
External links
| Identifiers: |
|---|
- Uroxatral (alfuzosin HCl) Extended-Release Tablets Prescribing Information Archived 2016-07-22 at the Wayback Machine
- Alfuzosin Archived 2016-07-05 at the Wayback Machine (General information from the NIH)