Carboplatin
 | |
 | |
| Names | |
|---|---|
| Pronunciation | /ˈkɑːrboʊˌplætən/ |
| Trade names | Paraplatin, others |
IUPAC name
| |
| Clinical data | |
| Drug class | Chemotherapy |
| Main uses | Ovarian cancer, lung cancer, head and neck cancer, brain cancer, neuroblastoma[1] |
| Side effects | Low blood cells, nausea, electrolyte problems[2][1] |
| WHO AWaRe | UnlinkedWikibase error: ⧼unlinkedwikibase-error-statements-entity-not-set⧽ |
| Pregnancy category |
|
| Routes of use | Intravenous |
| Defined daily dose | Not established[3] |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a695017 |
| Legal | |
| Legal status |
|
| Pharmacokinetics | |
| Bioavailability | complete |
| Protein binding | Very low |
| Elimination half-life | 1.1-2 hours |
| Excretion | Kidney |
| Chemical and physical data | |
| Formula | C6H12N2O4Pt |
| Molar mass | 371.256 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
Carboplatin, sold under the trade name Paraplatin among others, is a chemotherapy medication used to treat a number of forms of cancer.[1] This includes ovarian cancer, lung cancer, head and neck cancer, brain cancer, and neuroblastoma.[1] It is used by injection into a vein.[1]
Side effects generally occur.[1] Common side effects include low blood cell levels, nausea, and electrolyte problems.[2][1] Other serious side effects include allergic reactions and increased future risk of another cancer.[1] Use during pregnancy may result in harm to the baby.[1] Carboplatin is in the platinum-based antineoplastic family of medications and works by interfering with duplication of DNA.[1][4]
Carboplatin was patented in 1972 and approved for medical use in 1986.[5] It is on the World Health Organization's List of Essential Medicines.[6] The wholesale cost in the developing world is about US$26–38 per 450 mg vial.[7] In the United Kingdom it costs the National Health Service (NHS) about 160 pounds (around US$200).[8]
Medical uses
Carboplatin is used to treat a number of forms of cancer. This includes ovarian cancer, lung cancer, head and neck cancer, brain cancer, and neuroblastoma. It may be used for some types of testicular cancer but cisplatin is generally more effective.[1]
Dosage
The defined daily dose is not established[3]
Side-effects
Relative to cisplatin, the greatest benefit of carboplatin is its reduced side effects, particularly the elimination of nephrotoxic effects. Nausea and vomiting are less severe and more easily controlled.
The main drawback of carboplatin is its myelosuppressive effect. This causes the blood cell and platelet output of bone marrow in the body to decrease quite dramatically, sometimes as low as 10% of its usual production levels. The nadir of this myelosuppression usually occurs 21–28 days after the first treatment, after which the blood cell and platelet levels in the blood begin to stabilize, often coming close to its pre-carboplatin levels. This decrease in white blood cells (neutropenia) can cause complications, and is sometimes treated with drugs like filgrastim. The most notable complication of neutropenia is increased probability of infection by opportunistic organisms, which necessitates hospital readmission and treatment with antibiotics.
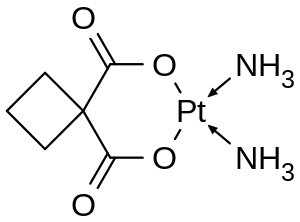
Chemistry
In terms of its structure, carboplatin differs from cisplatin in that it has a bidentate dicarboxylate (the ligand is CycloButane DiCarboxylic Acid, CBDCA) in place of the two chloride ligands, which are the leaving groups in cisplatin. For this reason, "CBDCA" is sometimes used in the medical literature as an abbreviation referring to carboplatin. Carboplatin exhibits lower reactivity and slower DNA binding kinetics, although it forms the same reaction products in vitro at equivalent doses with cisplatin. Unlike cisplatin, carboplatin may be susceptible to alternative mechanisms. Some results show that cisplatin and carboplatin cause different morphological changes in MCF-7 cell lines while exerting their cytotoxic behaviour.[9] The diminished reactivity limits protein-carboplatin complexes, which are excreted. The lower excretion rate of carboplatin means that more is retained in the body, and hence its effects are longer lasting (a retention half-life of 30 hours for carboplatin, compared to 1.5-3.6 hours in the case of cisplatin).
Mechanism of action
Two theories exist to explain the molecular mechanism of action of carboplatin with DNA:
- Aquation, or the like-cisplatin hypothesis.
- Activation, or the unlike-cisplatin hypothesis.
The former is more accepted owing to the similarity of the leaving groups with its predecessor cisplatin, while the latter hypothesis envisages a biological activation mechanism to release the active Pt2+ species.
Dose
Calvert's formula is used to calculate the dose of carboplatin. It takes under consideration the creatinine clearance and the desired area under curve.[10] After 24 hours, close to 70% of carboplatin is excreted in the urine unchanged. This means that the dose of carboplatin must be adjusted for any impairment in kidney function.[11]
Calvert formula:
The typical area under the curve (AUC) for carboplatin ranges from 3-7 (mg/ml)*min.[11]
History
Carboplatin was discovered at Michigan State University,[12] and developed at the Institute of Cancer Research in London. Bristol-Myers Squibb gained Food and Drug Administration (FDA) approval for carboplatin, under the brand name Paraplatin, in March 1989. Starting in October 2004, generic versions of the drug became available.
Research
Carboplatin has also been used for adjuvant therapy of stage 1 seminomatous testicular cancer. Research has indicated that it is not less effective than adjuvant radiotherapy for this treatment, while having fewer side effects.[13] This has led to carboplatin based adjuvant therapy being generally preferred over adjuvant radiotherapy in clinical practice.[14]
See also
References
- 1 2 3 4 5 6 7 8 9 10 11 "Carboplatin". The American Society of Health-System Pharmacists. Archived from the original on 21 December 2016. Retrieved 8 December 2016.
- 1 2 Oun R, Moussa YE, Wheate NJ (2018). "The side effects of platinum-based chemotherapy drugs: a review for chemists". Dalton Transactions. 47 (19): 6645–6653. doi:10.1039/c8dt00838h. PMID 29632935.
{{cite journal}}: CS1 maint: uses authors parameter (link) - 1 2 "WHOCC - ATC/DDD Index". www.whocc.no. Archived from the original on 26 September 2020. Retrieved 18 September 2020.
- ↑ Apps, M. G.; Choi, E. H. Y.; Wheate, N. J. (2015). "The state-of-play and future of platinum drugs". Endocrine-Related Cancer. 22 (4): 219–233. doi:10.1530/ERC-15-0237. PMID 26113607.
- ↑ Fischer, Janos; Ganellin, C. Robin (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 513. ISBN 9783527607495. Archived from the original on 2016-12-20.
- ↑ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ↑ "Carboplatin". International Drug Price Indicator Guide. Archived from the original on 22 January 2018. Retrieved 8 December 2016.
- ↑ British national formulary: BNF 69 (69 ed.). British Medical Association. 2015. p. 605. ISBN 9780857111562.
- ↑ Natarajan G, Malathi R, Holler E (November 1999). "Increased DNA-binding activity of cis-1,1-cyclobutanedicarboxylatodiammineplatinum(II) (carboplatin) in the presence of nucleophiles and human breast cancer MCF-7 cell cytoplasmic extracts: activation theory revisited". Biochemical Pharmacology. 58 (10): 1625–29. doi:10.1016/S0006-2952(99)00250-6. PMID 10535754.
- ↑ O'Cearbhaill, MD, Roisin; Sabbatini, MD, Paul S. (September 1, 2012). "New Guidelines for Carboplatin Dosing". Memorial Sloan Kettering Cancer Center. Archived from the original on 2014-10-31. Retrieved 2014-03-27.
- 1 2 Calvert, A H; Newell, D R; Gumbrell, L A; O'Reilly, S; Burnell, M; Boxall, F E; Siddik, Z H; Judson, I R; Gore, M E (1989-11-01). "Carboplatin dosage: prospective evaluation of a simple formula based on renal function". Journal of Clinical Oncology. 7 (11): 1748–1756. doi:10.1200/JCO.1989.7.11.1748. ISSN 0732-183X.
- ↑ "Discovery to Market: Fact Sheet". Archived from the original on 2012-02-08.
- ↑ Oliver RT, Mason MD, Mead GM, von der Maase H, Rustin GJ, Joffe JK, de Wit R, Aass N, Graham JD, Coleman R, Kirk SJ, Stenning SP (2005). "Radiotherapy versus single-dose carboplatin in adjuvant treatment of stage I seminoma: a randomised trial". Lancet. 366 (9482): 293–300. doi:10.1016/S0140-6736(05)66984-X. PMID 16039331.
- ↑ Toner GC (2015). "Testicular cancer: Optimal management of stage I seminoma in 2015". Nat Rev Urol. 12 (5): 249–51. doi:10.1038/nrurol.2015.85. PMID 25896179.
Additional references
- Canetta R, Rozencweig M, Carter SK (September 1985). "Carboplatin: the clinical spectrum to date". Cancer Treat. Rev. 12 (Suppl A): 125–36. doi:10.1016/0305-7372(85)90027-1. PMID 3002623.
- Yang XL, Wang AH (September 1999). "Structural studies of atom-specific anticancer drugs acting on DNA". Pharmacol. Ther. 83 (3): 181–215. doi:10.1016/S0163-7258(99)00020-0. PMID 10576292.
External links
| External sites: |
|
|---|---|
| Identifiers: |
- Creatinine Clearence [sic] Calculator Archived 2020-12-05 at the Wayback Machine