Dicloxacillin
 | |
| Names | |
|---|---|
| Pronunciation | dye klox' a sil' in[1] |
IUPAC name
| |
| Clinical data | |
| Drug class | Antibiotic (penicillin)[1] |
| Main uses | Staphylococcal infections[2] |
| Side effects | Nausea, diarrhea, rash, allergic reactions[1] |
| WHO AWaRe | UnlinkedWikibase error: ⧼unlinkedwikibase-error-statements-entity-not-set⧽ |
| Pregnancy category |
|
| Routes of use | By mouth |
| Typical dose | 125 to 500 mg QID[1] |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a685017 |
| Legal | |
| Legal status |
|
| Pharmacokinetics | |
| Bioavailability | 60 to 80% |
| Protein binding | 98% |
| Metabolism | Liver |
| Elimination half-life | 0.7 hours |
| Excretion | Kidney and biliary |
| Chemical and physical data | |
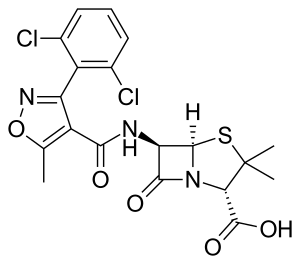
| Formula | C19H17Cl2N3O5S |
| Molar mass | 470.32 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
Dicloxacillin is an antibiotics used to treat Staphylococcal infections.[2] This may include cellulitis, osteomyelitis, and pneumonia.[3] While it is effective in penicillinase resistant disease it not effective for MRSA.[2] It is taken by mouth.[2]
Common side effects include nausea, diarrhea, rash, and allergic reactions.[1] Other side effects may include anaphylaxis, Clostridium difficile diarrhea and low white blood cells.[1] Use in pregnancy appears to be relatively safe, but has not been well studied.[4] It is a β-lactam antibiotic and penicillin.[1] It works by interfering with the bacterial cell wall.[1]
Dicloxacillin was patented in 1961 and approved for medical use in 1968.[5] It is available as a generic medication.[1] In the United States 40 pills of 500 mg costs about 30 USD as of 2021.[6]
Medical uses
Dicloxacillin is used to treat mild-to-moderate staphylococcal infections.[7] To decrease the development of resistance, dicloxacillin is recommended to treat infections that are suspected or proven to be caused by beta-lactamase-producing bacteria.[7]
Dicloxacillin is similar in pharmacokinetics, antibacterial activity, and indications to flucloxacillin, and the two agents are considered interchangeable.[8] It is believed to have lower incidence of severe live adverse effects than flucloxacillin, but a higher incidence of renal adverse effects.[8]
Dicloxacillin is used for the treatment of infections caused by susceptible bacteria. Specific approved indications include:[8]
- Staphylococcal skin infections and cellulitis – including impetigo, otitis externa, folliculitis, boils, carbuncles, and mastitis
- Pneumonia (adjunct)
- Osteomyelitis, septic arthritis, throat infections, streptococcus
- Septicaemia
- Empirical treatment for endocarditis
- Surgical prophylaxis
Dosage
It is typically taken at a dose of 125 to 500 mg every 6 hours.[1]
Dicloxacillin is commercially available as the sodium salt, dicloxacillin sodium, in capsules and as a powder for reconstitution.[9][10]
Contraindications
Dicloxacillin is contraindicated in those with a previous history of allergy (hypersensitivity/anaphylactic reaction) to any penicillins.[9][11]
Side effects
Common adverse drug reactions (ADRs) associated with the use of dicloxacillin include: diarrhoea, nausea, rash, urticaria, pain and inflammation at injection site, superinfection (including candidiasis), allergy, and transient increases in liver enzymes and bilirubin.[8]
On rare occasions, cholestatic jaundice (also referred to as cholestatic hepatitis) has been associated with dicloxacillin therapy. The reaction may occur up to several weeks after treatment has stopped, and takes weeks to resolve. The estimated incidence is 1 in 15,000 exposures, and is more frequent in people over 55 years old, females, and those with treatment longer than 2 weeks.[8]
It should be used with caution and monitored in the elderly, particularly with intravenous administration, due to a risk of thrombophlebitis.[9]
Dicloxacillin can also lower the effectiveness of birth control pills and pass into breast milk.[12]
Interactions
Dicloxacillin has potential interactions with following drugs:
Resistance
Despite dicloxacillin being insensitive to beta-lactamases, some organisms have developed resistance to other narrow-spectrum β-lactam antibiotics including methicillin. Such organisms include methicillin-resistant Staphylococcus aureus (MRSA).[16]
Mechanism of action
Like other β-lactam antibiotics, dicloxacillin acts by inhibiting the synthesis of bacterial cell walls. It inhibits cross-linkage between the linear peptidoglycan polymer chains that make up a major component of the cell wall of Gram-positive bacteria.
Medicinal chemistry
Dicloxacillin is insensitive to beta-lactamase (also known as penicillinase) enzymes secreted by many penicillin-resistant bacteria. The presence of the isoxazolyl group on the side chain of the penicillin nucleus facilitates the β-lactamase resistance, since they are relatively intolerant of side-chain steric hindrance. Thus, it is able to bind to penicillin-binding proteins (PBPs) and inhibit peptidoglycan crosslinking, but is not bound by or inactivated by β-lactamase
Society and culture
Dicloxacillin is available under a variety of trade names including Diclocil (BMS).[17]
See also
References
- 1 2 3 4 5 6 7 8 9 10 "Dicloxacillin". LiverTox: Clinical and Research Information on Drug-Induced Liver Injury. National Institute of Diabetes and Digestive and Kidney Diseases. 2012. Archived from the original on 28 August 2021. Retrieved 24 December 2021.
- 1 2 3 4 "Dicloxacillin Monograph for Professionals". Drugs.com. Archived from the original on 20 January 2021. Retrieved 24 December 2021.
- ↑ "289107 DICLOXACILLIN MYLAN 500 dicloxacillin (as sodium) 500 mg capsule bottle". Archived from the original on 11 January 2022. Retrieved 24 December 2021.
- ↑ "Dicloxacillin Use During Pregnancy". Drugs.com. Archived from the original on 3 December 2020. Retrieved 24 December 2021.
- ↑ Fischer J, Ganellin CR (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 491. ISBN 9783527607495. Archived from the original on 2021-05-20. Retrieved 2020-12-01.
- ↑ "Dicloxacillin Prices, Coupons & Savings Tips - GoodRx". GoodRx. Archived from the original on 6 February 2017. Retrieved 24 December 2021.
- 1 2 Dicloxacillin. LiverTox: Clinical and Research Information on Drug-Induced Liver Injury [Internet]. Bethesda (MD): National Institute of Diabetes and Digestive and Kidney Diseases. 2012. Archived from the original on 2021-08-28. Retrieved 2020-12-01.
- 1 2 3 4 5 Rossi S, editor. Australian Medicines Handbook 2006. Adelaide: Australian Medicines Handbook; 2006
- 1 2 3 Product Information: DICLOXACILLIN SODIUM-dicloxacillin sodium capsule. Teva Pharmaceuticals USA Inc, Revised 8/2015
- ↑ "Dicloxacillin". MedlinePlus Drug Information. U.S. National Library of Medicine, Department of Health and Human Services, National Institutes of Health. Archived from the original on 2020-10-23. Retrieved 2020-12-01.
- ↑ "DICLOXACILLIN SODIUM- dicloxacillin sodium capsule". DailyMed. National Institutes of Health, U.S. National Library of Medicine, Health & Human Services. Archived from the original on 2020-11-30. Retrieved 2020-12-01.
- ↑ "Dicloxacillin - Side Effects, Dosage, Interactions - Drugs - Everyday Health". EverydayHealth.com. Archived from the original on 2020-10-22. Retrieved 2020-12-01.
- ↑ Lacey CS (May 2004). "Interaction of dicloxacillin with warfarin". The Annals of Pharmacotherapy. 38 (5): 898. doi:10.1345/aph.1d484. PMID 15054148. S2CID 19182647.
- ↑ Ronchera CL, Hernández T, Peris JE, Torres F, Granero L, Jiménez NV, Plá JM (October 1993). "Pharmacokinetic interaction between high-dose methotrexate and amoxycillin". Therapeutic Drug Monitoring. 15 (5): 375–9. doi:10.1097/00007691-199310000-00004. PMID 8249043. S2CID 28111642.
- ↑ Moellering RC (August 1983). "Rationale for use of antimicrobial combinations". The American Journal of Medicine. 75 (2A): 4–8. doi:10.1016/0002-9343(83)90088-8. PMID 6351605.
- ↑ Rosdahl VT, Frimodt-Møller N, Bentzon MW (August 1989). "Resistance to dicloxacillin, methicillin and oxacillin in methicillin-susceptible and methicillin-resistant Staphylococcus aureus detected by dilution and diffusion methods". APMIS. 97 (8): 715–22. doi:10.1111/j.1699-0463.1989.tb00467.x. PMID 2669854. S2CID 29646870.
- ↑ Miranda-Novales G, Leaños-Miranda BE, Vilchis-Pérez M, Solórzano-Santos F (October 2006). "In vitro activity effects of combinations of cephalothin, dicloxacillin, imipenem, vancomycin and amikacin against methicillin-resistant Staphylococcus spp. strains". Annals of Clinical Microbiology and Antimicrobials. 5: 25. doi:10.1186/1476-0711-5-25. PMC 1617116. PMID 17034644.
External links
| Identifiers: |
|---|