25E-NBOH
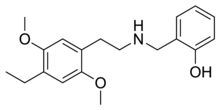
25E-NBOH (2C-E-NBOH, NBOH-2C-E) is a derivative of the phenethylamine derived hallucinogen 2C-E. It was first developed by Martin Hansen at the University of Copenhagen in 2010 as a brain imaging agent,[2] but has subsequently been sold as a designer drug, first being identified in Brazil in 2018 on seized blotter paper,[3][4][5] as well as in Slovenia.[6] It acts as a potent serotonin receptor agonist with similar affinity to better-known compounds such as 25I-NBOMe at 5-HT2A and 5-HT2C receptors.[7]
 | |
| Legal status | |
|---|---|
| Legal status | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| UNII | |
| Chemical and physical data | |
| Formula | C19H25NO3 |
| Molar mass | 315.413 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Legal status
United Kingdom
This substance is a Class A drug in the United Kingdom as a result of the N-benzylphenethylamine catch-all clause in the Misuse of Drugs Act 1971.[8]
See also
References
- Anvisa (2023-07-24). "RDC Nº 804 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 804 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 2023-07-25). Archived from the original on 2023-08-27. Retrieved 2023-08-27.
- Hansen M (2010-12-16). Design and Synthesis of Selective Serotonin Receptor Agonists for Positron Emission Tomography Imaging of the Brain (Ph.D. thesis). University of Copenhagen. doi:10.13140/RG.2.2.33671.14245.
- Machado Y, Neto JC, Lordeiro RA, Silva MF, Piccin E (January 2019). "Profile of new psychoactive substances (NPS) and other synthetic drugs in seized materials analysed in a Brazilian forensic laboratory". Forensic Toxicology. 37 (1): 265–71. doi:10.1007/s11419-018-0456-3. S2CID 53812909.
- Machado Y, Neto JC, Lordeiro RA, Alves RB, Piccin E (January 2020). "Identification of new NBOH drugs in seized blotter papers: 25B-NBOH, 25C-NBOH, and 25E-NBOH". Forensic Toxicology. 38 (1): 203–15. doi:10.1007/s11419-019-00509-7. S2CID 209672508.
- Rodrigues de Morais D, Francisco da Cunha K, Betoni Rodrigues T, Lanaro R, de Melo Barbosa L, Jardim Zacca J, Nogueira Eberlin M, Costa JL (April 2020). "Triple quadrupole-mass spectrometry protocols for the analysis of NBOMes and NBOHs in blotter papers". Forensic Science International. 309: 110184. doi:10.1016/j.forsciint.2020.110184. PMID 32086005. S2CID 211246833.
- Analytical Report. 25E-NBOH. Nacionalni Forenzični Laboratorij, Slovenia, 24 January 2018
- Hansen M, Phonekeo K, Paine JS, Leth-Petersen S, Begtrup M, Bräuner-Osborne H, Kristensen JL (March 2014). "Synthesis and structure-activity relationships of N-benzyl phenethylamines as 5-HT2A/2C agonists". ACS Chemical Neuroscience. 5 (3): 243–9. doi:10.1021/cn400216u. PMC 3963123. PMID 24397362.
- "The Misuse of Drugs Act 1971 (Ketamine etc.) (Amendment) Order 2014". www.legislation.gov.uk.
| 5-HT1 |
| ||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 5-HT2 |
| ||||||||||||||||||||||||||||||||||||||
| 5-HT3–7 |
| ||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||
| Phenethylamines |
|
|---|---|
| Amphetamines |
|
| Phentermines |
|
| Cathinones |
|
| Phenylisobutylamines | |
| Phenylalkylpyrrolidines | |
| Catecholamines (and close relatives) |
|
| Miscellaneous |
|
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.