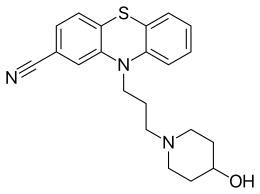
Periciazine
Periciazine (INN), also known as pericyazine (BAN) or propericiazine, is a drug that belongs to the phenothiazine class of typical antipsychotics.
 | |
| Clinical data | |
|---|---|
| Pregnancy category |
|
| Routes of administration | Oral |
| Drug class | Typical antipsychotic |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Metabolism | Hepatic (mostly via conjugation)[2] |
| Elimination half-life | 12 h[2] |
| Excretion | Renal[2] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank |
|
| ChemSpider |
|
| UNII | |
| KEGG |
|
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.018.248 |
| Chemical and physical data | |
| Formula | C21H23N3OS |
| Molar mass | 365.50 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Periciazine is not approved for sale in the United States. It is commonly sold in Canada, Italy and Russia under the tradename Neuleptil and in United Kingdom and Australia under the tradename Neulactil.[3]
Medical uses
The primary uses of periciazine include in the short-term treatment of severe anxiety or tension and in the maintenance treatment of psychotic disorders such as schizophrenia. There is insufficient evidence to determine whether periciazine is more or less effective than other antipsychotics.[2] A 2014 systematic review compared periciazine with typical antipsychotics for schizophrenia:
| Summary | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| On the basis of very low quality evidence it is not possible to determine the effects of periciazine in comparison with antipsychotics such as chlorpromazine or trifluoperazine for the treatment of schizophrenia. There is some evidence, however, that periciazine may be associated with a higher incidence of extrapyramidal side effects than other antipsychotics.[4] | ||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||
Periciazine has also been studied in the treatment of opioid dependence.[5]
Adverse effects
Periciazine is a rather sedating and anticholinergic antipsychotic, and despite being classed with the typical antipsychotics, its risk of extrapyramidal side effects is comparatively low.[6] It has a relatively high risk of causing hyperprolactinaemia and a moderate risk of causing weight gain and orthostatic hypotension.[6]
Synthesis
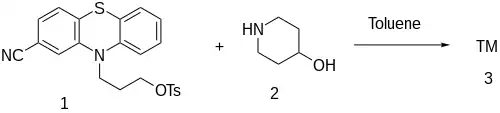
The final step in the synthesis involves the alkylation of 3-(2-cyanophenothiazin-10-yl)propyl 4-methylbenzenesulfonate, CID:134990672 (1) with 4-Piperidinol [5382-16-1] (2) giving Periciazine (3).
References
- Anvisa (2023-03-31). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 2023-04-04). Archived from the original on 2023-08-03. Retrieved 2023-08-16.
- "NEULACTIL PRODUCT INFORMATION" (PDF). TGA eBusiness Services. sanofi-aventis australia pty ltd. 13 September 2011. Retrieved 2 November 2013.
- Pericyazine. 23 September 2011. Retrieved 2 November 2013.
{{cite book}}:|work=ignored (help) - Matar HE, Almerie MQ, Makhoul S, Xia J, Humphreys P (May 2014). "Pericyazine for schizophrenia". The Cochrane Database of Systematic Reviews. 5 (5): CD007479. doi:10.1002/14651858.CD007479.pub2. PMID 24825770.
- Sivolap IuP, Savchenkov VA (1999). "[The use of neuroleptics in treating opiate dependence]". Zhurnal Nevrologii I Psikhiatrii Imeni S.S. Korsakova (in Russian). 99 (6): 29–34. PMID 10441864.
- "Approximate relative frequency (not intensity) of common adverse effects of antipsychotics (Table 8.21) [NB1]". eTherapeutic Guidelines complete. Therapeutic Guidelines Limited. February 2013. Retrieved 2 November 2013.
- FR1212031 idem Robert Jacques Georges, Jacob Robert Michel, U.S. Patent 3,075,976 (1963 to Rhone Poulenc Sa).
External links
- Neulactil - Summary of Product Characteristics from the electronic Medicines Compendium
- from the Therapeutic Goods Administration.
- from Health Canada.
| Classes | |
|---|---|
| Antidepressants (Tricyclic antidepressants (TCAs)) |
|
| Antihistamines |
|
| Antipsychotics |
|
| Anticonvulsants | |
| Anticholinergics | |
| Others |
|