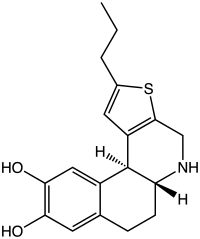
A-86929
 | |
| Names | |
|---|---|
| Preferred IUPAC name
(5aR,11bS)-2-Propyl-4,5,5a,6,7,11b-hexahydrobenzo[f]thieno[2,3-c]quinoline-9,10-diol | |
| Identifiers | |
CAS Number |
|
3D model (JSmol) |
|
| ChEMBL | |
| ChemSpider | |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula |
C18H21NO2S |
| Molar mass | 315.429 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
A-86929 is a synthetic compound that acts as a selective dopamine receptor D1 agonist.[1][2] It was developed as a possible treatment for Parkinson's disease,[3] as well as for other applications such as treatment of cocaine addiction,[4][5] but while it had reasonable efficacy in humans it also caused dyskinesias and has not been continued.[6][7] It has mainly been used as its diacetate ester prodrug adrogolide (ABT-431), which has better bioavailability.[8][9]
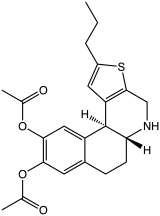
Adrogolide
References
- ↑ Michaelides MR, Hong Y, DiDomenico S, Asin KE, Britton DR, Lin CW, Williams M, Shiosaki K (September 1995). "(5aR,11bS)-4,5,5a,6,7,11b-hexahydro-2-propyl-3-thia-5-azacyclopent-1- ena[c]-phenanthrene-9,10-diol (A-86929): a potent and selective dopamine D1 agonist that maintains behavioral efficacy following repeated administration and characterization of its diacetyl prodrug (ABT-431)". Journal of Medicinal Chemistry. 38 (18): 3445–7. doi:10.1021/jm00018a002. PMID 7658429.
- ↑ Ehrlich PP, Ralston JW, Michaelides MR (May 1997). "An Efficient Enantioselective Synthesis of the D1 Agonist (5aR,11bS)-4,5,5a,6,7,11b-Hexahydro-2-propyl-3-thia- 5-azacyclopenta[c]phenanthrene-9,10-diol (A-86929)". The Journal of Organic Chemistry. 62 (9): 2782–2785. doi:10.1021/jo970066l. PMID 11671640.
- ↑ Rascol O, Blin O, Thalamas C, Descombes S, Soubrouillard C, Azulay P, Fabre N, Viallet F, Lafnitzegger K, Wright S, Carter JH, Nutt JG (June 1999). "ABT-431, a D1 receptor agonist prodrug, has efficacy in Parkinson's disease". Annals of Neurology. 45 (6): 736–41. doi:10.1002/1531-8249(199906)45:6<736::AID-ANA7>3.0.CO;2-F. PMID 10360765.
- ↑ Haney M, Collins ED, Ward AS, Foltin RW, Fischman MW (March 1999). "Effect of a selective dopamine D1 agonist (ABT-431) on smoked cocaine self-administration in humans". Psychopharmacology. 143 (1): 102–10. doi:10.1007/s002130050925. PMID 10227086.
- ↑ Gorelick DA, Gardner EL, Xi ZX (2004). "Agents in development for the management of cocaine abuse". Drugs. 64 (14): 1547–73. doi:10.2165/00003495-200464140-00004. PMID 15233592.
- ↑ Rascol O, Nutt JG, Blin O, Goetz CG, Trugman JM, Soubrouillard C, Carter JH, Currie LJ, Fabre N, Thalamas C, Giardina WW, Wright S (February 2001). "Induction by dopamine D1 receptor agonist ABT-431 of dyskinesia similar to levodopa in patients with Parkinson disease". Archives of Neurology. 58 (2): 249–54. doi:10.1001/archneur.58.2.249. PMID 11176963.
- ↑ Zhang J, Xiong B, Zhen X, Zhang A (March 2009). "Dopamine D1 receptor ligands: where are we now and where are we going". Medicinal Research Reviews. 29 (2): 272–94. doi:10.1002/med.20130. PMID 18642350.
- ↑ Shiosaki K, Jenner P, Asin KE, Britton DR, Lin CW, Michaelides M, Smith L, Bianchi B, Didomenico S, Hodges L, Hong Y, Mahan L, Mikusa J, Miller T, Nikkel A, Stashko M, Witte D, Williams M (January 1996). "ABT-431: the diacetyl prodrug of A-86929, a potent and selective dopamine D1 receptor agonist: in vitro characterization and effects in animal models of Parkinson's disease". The Journal of Pharmacology and Experimental Therapeutics. 276 (1): 150–60. PMID 8558425.
- ↑ Giardina WJ, Williams M (2001). "Adrogolide HCl (ABT-431; DAS-431), a prodrug of the dopamine D1 receptor agonist, A-86929: preclinical pharmacology and clinical data". CNS Drug Reviews. 7 (3): 305–16. doi:10.1111/j.1527-3458.2001.tb00201.x. PMC 6741696. PMID 11607045.
This article is issued from Offline. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.