Flumetasone
 | |
| Clinical data | |
|---|---|
| Other names | Flumethasone (USAN) |
| AHFS/Drugs.com | Micromedex Detailed Consumer Information |
| Routes of administration | Topical |
| ATC code | |
| Pharmacokinetic data | |
| Metabolism | Hepatic, CYP3A4-mediated |
| Identifiers | |
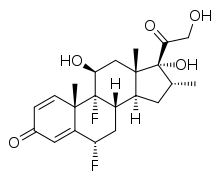
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.016.701 |
| Chemical and physical data | |
| Formula | C22H28F2O5 |
| Molar mass | 410.458 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
| | |
Flumetasone, also known as flumethasone,[1] is a corticosteroid for topical use.
It was patented in 1951 and approved for medical use in 1964.[2]
Chemistry
Flumethasone is 420 times as potent as cortisone in an animal model for anti-inflammatory activity.
Names
Trade names include Locacorten, Locorten, and Orsalin. It is available in combination with clioquinol, under the trade name Locacorten-Vioform (in some countries Locorten-Vioform), for the treatment of otitis externa and otomycosis. It is usually formulated as the pivalic acid ester prodrug called flumetasone pivalate.
See also
References
- ↑ Morton IK, Hall JM (1999). Concise Dictionary of Pharmacological Agents Properties and Synonyms. Dordrecht: Springer Netherlands. p. 122. ISBN 9789401144391.
- ↑ Fischer J, Ganellin CR (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 484. ISBN 9783527607495.
This article is issued from Offline. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.