Α-PHiP
 | |
| Legal status | |
|---|---|
| Legal status |
|
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| Chemical and physical data | |
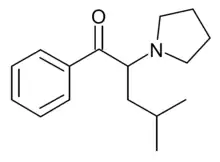
| Formula | C16H23NO |
| Molar mass | 245.366 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
α-PHiP (also known as α-PiHP) is a stimulant drug of the cathinone class that has been sold online as a designer drug. It is a positional isomer of pyrovalerone, with the methyl group shifted from the 4-position of the aromatic ring to the 4-position of the acyl chain. In a classic 2006 study of pyrrolidinyl cathinone derivatives by Meltzer et al. at Organix, the alpha-isobutyl derivative of pyrovalerone, O-2494, was found to have the highest potency in vitro as an inhibitor of the dopamine transporter of the alpha substituted derivatives tested;[2] however, it was not until ten years later in July 2016 that α-PHiP was first identified as a designer drug,[3] when it was reported to the EMCDDA by a forensic laboratory in Slovenia.[4][5]
See also
References
- ↑ Anvisa (2023-07-24). "RDC Nº 804 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 804 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 2023-07-25). Archived from the original on 2023-08-27. Retrieved 2023-08-27.
- ↑ Meltzer PC, Butler D, Deschamps JR, Madras BK (February 2006). "1-(4-Methylphenyl)-2-pyrrolidin-1-yl-pentan-1-one (Pyrovalerone) analogues: a promising class of monoamine uptake inhibitors". Journal of Medicinal Chemistry. 49 (4): 1420–32. doi:10.1021/jm050797a. PMC 2602954. PMID 16480278.
- ↑ "Analytic Report alpha-PHiP (C16H23NO) 4-methyl-1-phenyl-2-(pyrrolidin-1-yl)pentan-1-one" (PDF). Slovenia: Nacionalni forenzični laboratorij. July 2016. Archived (PDF) from the original on 2020-10-03. Retrieved 2019-10-24.
- ↑ "Europol 2016 Annual Report on the implementation of Council Decision 2005/387/JHA" (PDF). EMCDDA–Europol Joint Publication. Archived (PDF) from the original on 2021-01-26. Retrieved 2019-10-24.
- ↑ Liu C, Jia W, Li T, Hua Z, Qian Z (August 2017). "Identification and analytical characterization of nine synthetic cathinone derivatives N-ethylhexedrone, 4-Cl-pentedrone, 4-Cl-α-EAPP, propylone, N-ethylnorpentylone, 6-MeO-bk-MDMA, α-PiHP, 4-Cl-α-PHP, and 4-F-α-PHP". Drug Testing and Analysis. 9 (8): 1162–1171. doi:10.1002/dta.2136. PMID 27863142.
This article is issued from Offline. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.