Furethidine
 | |
| Clinical data | |
|---|---|
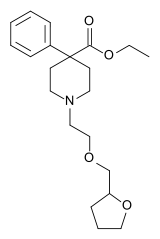
| Other names | ethyl 4-phenyl-1-(2-tetrahydrofurfuryloxyethyl)piperidine-4-carboxylate |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| ECHA InfoCard | 100.017.451 |
| Chemical and physical data | |
| Formula | C21H31NO4 |
| Molar mass | 361.482 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
| (verify) | |
Furethidine[1] is a 4-phenylpiperidine derivative that is related to the clinically used opioid analgesic drug pethidine (meperidine),[2] but with around 25x higher potency.[3]
Furethidine is not currently used in medicine and is a Class A/Schedule I drug which is controlled under UN drug conventions. It has similar effects to other opioid derivatives, such as analgesia, sedation, nausea and respiratory depression.[4] In the United States it is a Schedule I Narcotic controlled substance with the ACSCN of 9626.[5]
References
- ↑ GB 797448, Frearson PM, Stern ES, "Novel piperidine compounds and their production", published 2 July 1958, assigned to J F Macfarlan & Co Ltd
- ↑ Maul C, Buschmann H, Sundermann B (2005). "Opioids: 3.3 Synthetic Opioids.". Analgesics. pp. 159–169. ISBN 978-3-527-30403-5.
- ↑ Casy AF, Parfitt RY. Opioid analgesics, chemistry and receptors. 1986, Plenum Press, New York. pp 234-235. ISBN 0-306-42130-5
- ↑ Cahal DA, Dare JG, Keith D (February 1961). "A sequential trial of analgesics in labour". The Journal of Obstetrics and Gynaecology of the British Commonwealth. 68: 88–93. doi:10.1111/j.1471-0528.1961.tb02689.x. PMID 13689779. S2CID 27397119.
- ↑ "Controlled Substances - Alphabetical Order" (PDF). Diversion Control Division. Drug Enforcement Agency, U.S. Department of Justice. 2020. p. 9.
External links
This article is issued from Offline. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.