Laudanosine
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
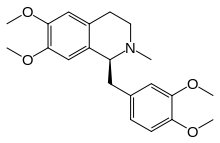
(1S)-1-[(3,4-Dimethoxyphenyl)methyl]-6,7-dimethoxy-2-methyl-1,2,3,4-tetrahydroisoquinoline | |
| Other names
N-Methyl-1,2,3,4-tetrahydropapaverine | |
| Identifiers | |
CAS Number |
|
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.018.412 |
| EC Number |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula |
C21H27NO4 |
| Molar mass | 357.450 g·mol−1 |
| Melting point | 89 °C (192 °F; 362 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Laudanosine or N-methyltetrahydropapaverine is a recognized metabolite[1] of atracurium and cisatracurium. Laudanosine decreases the seizure threshold, and thus it can induce seizures if present at sufficient threshold concentrations; however such concentrations are unlikely to be produced consequent to chemodegradable metabolism of clinically administered doses of cisatracurium or atracurium.

Laudanosine also occurs naturally in minute amounts (0.1%) in opium, from which it was first isolated in 1871.[2] Partial dehydrogenation of laudanosine will lead to papaverine, the alkaloid found in the opium poppy plant (Papaver somniferum).
Laudanosine is a benzyltetrahydroisoquinoline alkaloid. It has been shown to interact with GABA receptors, glycine receptors, opioid receptors, and nicotinic acetylcholine receptors,[1][3][4] but not benzodiazepine or muscarinic receptors, which are also involved in epilepsy and other types of seizures.[5]
References
- 1 2 Fodale V, Santamaria LB (July 2002). "Laudanosine, an atracurium and cisatracurium metabolite". Eur J Anaesthesiol. 19 (7): 466–73. doi:10.1017/s0265021502000777. PMID 12113608.
- ↑ Burger A (2005) [1954]. "The Benzylisoquinoline Alkaloids". In Manske RH, Holmes HL (eds.). The Alkaloids: Chemistry and Physiology. Vol. 4. New York: Academic Press. p. 48. ISBN 0-12-469504-3. Retrieved September 18, 2008 through Google Book Search.
- ↑ Katz Y, Weizman A, Pick CG, Pasternak GW, Liu L, Fonia O, Gavish M (May 1994). "Interactions between laudanosine, GABA, and opioid subtype receptors: implication for laudanosine seizure activity". Brain Res. 646 (2): 235–241. doi:10.1016/0006-8993(94)90084-1. PMID 8069669. S2CID 35031924.
- ↑ Exley R, Iturriaga-Vásquez P, Lukas RJ, Sher E, Cassels BK, Bermudez I (Sep 2005). "Evaluation of benzyltetrahydroisoquinolines as ligands for neuronal nicotinic acetylcholine receptors". Br J Pharmacol. 146 (1): 15–24. doi:10.1038/sj.bjp.0706307. PMC 1576253. PMID 15980871.
- ↑ Katz Y, Gavish M (Jan 1989). "Laudanosine does not displace receptor-specific ligands from the benzodiazepinergic or muscarinic receptors". Anesthesiology. 70 (1): 109–111. doi:10.1097/00000542-198901000-00020. PMID 2536252.