Normorphine
 | |
| Clinical data | |
|---|---|
| Other names | Normorphine |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.006.712 |
| Chemical and physical data | |
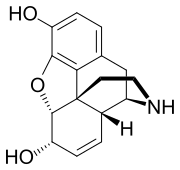
| Formula | C16H17NO3 |
| Molar mass | 271.316 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
| | |
Normorphine is an opiate analogue, the N-demethylated derivative of morphine, that was first described in the 1950s[1] when a large group of N-substituted morphine analogues were characterized for activity. The compound has relatively little opioid activity in its own right,[2][3] but is a useful intermediate which can be used to produce both opioid antagonists such as nalorphine, and also potent opioid agonists such as N-phenethylnormorphine.[4] with its formation from morphine catalyzed by the liver enzymes CYP3A4 and CYP2C8.[5]
Normorphine is a controlled substance listed under the Single Convention On Narcotic Drugs 1961 and the laws in various states implementing it; for example, in the United States it is a Schedule I Narcotic controlled substance, with an ACSCN of 9313 and an annual aggregate manufacturing quota of 18 grams in 2014, unchanged from the prior year. The salts in use are the free base hexahydrate (free base conversion ratio 0.715), and hydrochloride (0.833).[6]
References
- ↑ Clark RL, Pessolano AA, Weijlard J, Pfister K (October 1953). "N-Substituted epoxymorphinans". Journal of the American Chemical Society. 75 (20): 4963–7. doi:10.1021/ja01116a024.
- ↑ Fraser HF, Wikler A, Van Horn GD, Eisenman AJ, Isbell H (March 1958). "Human pharmacology and addiction liability of normorphine". The Journal of Pharmacology and Experimental Therapeutics. 122 (3): 359–69. PMID 13539761.
- ↑ Lasagna L, De Kornfeld TJ (November 1958). "Analgesic potency of normorphine in patients with postoperative pain". The Journal of Pharmacology and Experimental Therapeutics. 124 (3): 260–3. PMID 13588540.
- ↑ Yeh SY (January 1975). "Urinary excretion of morphine and its metabolites in morphine-dependent subjects". The Journal of Pharmacology and Experimental Therapeutics. 192 (1): 201–10. PMID 235634.
- ↑ Projean D, Morin PE, Tu TM, Ducharme J (August 2003). "Identification of CYP3A4 and CYP2C8 as the major cytochrome P450 s responsible for morphine N-demethylation in human liver microsomes". Xenobiotica; the Fate of Foreign Compounds in Biological Systems. 33 (8): 841–54. doi:10.1080/0049825031000121608. PMID 12936704. S2CID 41467595.
- ↑ "Quotas - 2014". DEA Diversion Control Division.