Ro60-0175
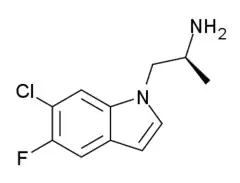 | |
| Identifiers | |
|---|---|
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| ChemSpider | |
| ChEBI | |
| ECHA InfoCard | 100.189.524 |
| Chemical and physical data | |
| Formula | C11H12ClFN2 |
| Molar mass | 226.68 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
| (verify) | |
Ro60-0175 is a drug developed by Hoffmann–La Roche, which has applications in scientific research.[1][2] It acts as a potent and selective agonist for both the 5-HT2B and 5-HT2C serotonin receptor subtypes, with good selectivity over the closely related 5-HT2A subtype, and little or no affinity at other receptors.[3][4]
See also
References
- ↑ Quarta D, Naylor CG, Stolerman IP (August 2007). "The serotonin 2C receptor agonist Ro-60-0175 attenuates effects of nicotine in the five-choice serial reaction time task and in drug discrimination". Psychopharmacology. 193 (3): 391–402. doi:10.1007/s00213-007-0802-3. PMID 17473916. S2CID 21020653.
- ↑ Fletcher PJ, Rizos Z, Sinyard J, Tampakeras M, Higgins GA (May 2008). "The 5-HT2C receptor agonist Ro60-0175 reduces cocaine self-administration and reinstatement induced by the stressor yohimbine, and contextual cues". Neuropsychopharmacology. 33 (6): 1402–12. doi:10.1038/sj.npp.1301509. PMID 17653111.
- ↑ Porter RH, Benwell KR, Lamb H, Malcolm CS, Allen NH, Revell DF, et al. (September 1999). "Functional characterization of agonists at recombinant human 5-HT2A, 5-HT2B and 5-HT2C receptors in CHO-K1 cells". British Journal of Pharmacology. 128 (1): 13–20. doi:10.1038/sj.bjp.0702751. PMC 1571597. PMID 10498829.
- ↑ Damjanoska KJ, Muma NA, Zhang Y, D'Souza DN, Garcia F, Carrasco GA, et al. (March 2003). "Neuroendocrine evidence that (S)-2-(chloro-5-fluoro-indol- l-yl)-1-methylethylamine fumarate (Ro 60-0175) is not a selective 5-hydroxytryptamine(2C) receptor agonist". The Journal of Pharmacology and Experimental Therapeutics. 304 (3): 1209–16. doi:10.1124/jpet.102.043489. PMID 12604698. S2CID 23880629.
This article is issued from Offline. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.