Bay R 1531
 | |
| Identifiers | |
|---|---|
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
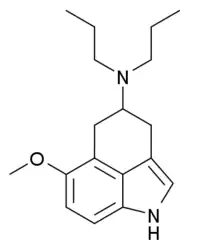
| Formula | C18H28N2O |
| Molar mass | 288.435 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
Bay R 1531 is a tricyclic tryptamine derivative which acts as a selective serotonin receptor 5-HT1A agonist. It was researched unsuccessfully for the treatment of stroke but remains in use for scientific research.[1][2][3][4]
See also
References
- ↑ Bielenberg GW, Burkhardt M (December 1990). "5-hydroxytryptamine1A agonists. A new therapeutic principle for stroke treatment". Stroke. 21 (12 Suppl): IV161-3. PMID 2148035.
- ↑ Critchley MA, Njung'e K, Handley SL (1992). "Actions and some interactions of 5-HT1A ligands in the elevated X-maze and effects of dorsal raphe lesions". Psychopharmacology. 106 (4): 484–90. doi:10.1007/bf02244819. PMID 1533721. S2CID 8404850.
- ↑ Canto de Souza A, Nunes de Souza RL, Péla IR, Graeff FG (March 1997). "High intensity social conflict in the Swiss albino mouse induces analgesia modulated by 5-HT1A receptors". Pharmacology, Biochemistry, and Behavior. 56 (3): 481–6. doi:10.1016/s0091-3057(96)00246-8. PMID 9077586. S2CID 20078255.
- ↑ Canto-de-Souza A, Nunes de Souza RL, Pelá IR, Graeff FG (March 1998). "Involvement of the midbrain periaqueductal gray 5-HT1A receptors in social conflict induced analgesia in mice". European Journal of Pharmacology. 345 (3): 253–6. doi:10.1016/s0014-2999(98)00018-1. PMID 9592023.
|
This article is issued from Offline. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.