Capsaicin
 | |
 | |
| Names | |
|---|---|
| Pronunciation | /kæpˈseɪsɪn/ or /kæpˈseɪəsɪn/ |
| Preferred IUPAC name
(6E)-N-[(4-Hydroxy-3-methoxyphenyl)methyl]-8-methylnon-6-enamide | |
| Other names
(E)-N-(4-Hydroxy-3-methoxybenzyl)-8-methylnon-6-enamide 8-Methyl-N-vanillyl-trans-6-nonenamide trans-8-Methyl-N-vanillylnon-6-enamide (E)-Capsaicin Capsicine Capsicin CPS | |
| Identifiers | |
CAS Number |
|
3D model (JSmol) |
|
Beilstein Reference |
2816484 |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| EC Number |
|
| KEGG | |
PubChem CID |
|
| UNII | |
InChI
| |
SMILES
| |
| Properties | |
Chemical formula |
C18H27NO3 |
| Molar mass | 305.418 g·mol−1 |
| Appearance | Crystalline white powder[1] |
| Odor | Highly volatile and pungent |
| Melting point | 62 to 65 °C (144 to 149 °F; 335 to 338 K) |
| Boiling point | 210 to 220 °C (410 to 428 °F; 483 to 493 K) 0.01 Torr |
Solubility in water |
0.0013 g/100 mL |
| Solubility | |
| Vapor pressure | 1.32×10−8 mm Hg at 25 °C[2] |
| UV-vis (λmax) | 280 nm |
| Structure | |
Crystal structure |
Monoclinic |
| Pharmacology | |
| M02AB01 (WHO) N01BX04 (WHO) | |
| License data | |
| Hazards | |
| Safety data sheet | [2] |
| GHS pictograms |    |
| GHS Signal word | Danger |
GHS hazard statements |
H301, H302, H315, H318 |
GHS precautionary statements |
P264, P270, P280, P301+310, P301+312, P302+352, P305+351+338, P310, P321, P330, P332+313, P362, P405, P501 |
| NFPA 704 (fire diamond) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
| Capsaicin | |
|---|---|
| Heat | Above peak (pure Capsaicin is toxic)[2] |
| Scoville scale | 16,000,000[3] SHU |
Capsaicin (8-methyl-N-vanillyl-6-nonenamide) (/kæpˈseɪsɪn/ or /kæpˈseɪəsɪn/) is an active component of chili peppers, which are plants belonging to the genus Capsicum. It is a chemical irritant for mammals, including humans, and produces a sensation of burning in any tissue with which it comes into contact. Capsaicin and several related alkaloids are called capsaicinoids and are produced as secondary metabolites by chili peppers, probably as deterrents against certain mammals and fungi.[4] Pure capsaicin is a hydrophobic, colorless, highly pungent,[2] crystalline to waxy solid compound.
Uses
Food

Because of the burning sensation caused by capsaicin when it comes in contact with mucous membranes, it is commonly used in food products to provide added spiciness or "heat" (piquancy), usually in the form of spices such as chili powder and paprika.[5] In high concentrations, capsaicin will also cause a burning effect on other sensitive areas, such as skin or eyes.[6] The degree of heat found within a food is often measured on the Scoville scale.[5]
There has long been a demand for capsaicin-spiced products like chili pepper, and hot sauces such as Tabasco sauce and Mexican salsa.[5] It is common for people to experience pleasurable and even euphoric effects from ingesting capsaicin.[5] Folklore among self-described "chiliheads" attribute this to pain-stimulated release of endorphins, a different mechanism from the local receptor overload that makes capsaicin effective as a topical analgesic.[6]
Medical use

Capsaicin is used as a pain medication in topical ointments and dermal patches, typically in concentrations between 0.025% and 0.1%.[7] Evidence of benefit in neuropathic pain and osteoarthritis; however is unclear as of 2018.[8] It may be applied for minor aches and pains of muscles and joints associated with arthritis, backache, strains and sprains, often in compounds with other rubefacients.[7] It is also used in peripheral neuropathy, such as post-herpetic neuralgia caused by shingles.[7] A capsaicin transdermal patch for pain due to post-herpetic neuralgia was approved in 2009 in the United States.[9][10] and the European Union.[11] Use for HIV neuralgia was declined.[12] One 2017 review found that high-dose topical capsaicin (8%) compared with 0.4% capsaicin provided moderate to substantial pain relief from post-herpetic neuralgia, HIV-neuropathy, and diabetic neuropathy.[13]
Although capsaicin creams have been used to treat psoriasis for reduction of itching,[7][14][15] a review of topical capsaicin for treatment of pruritus concluded there was insufficient evidence of effect.[16] Capsaicin decreases LDL cholesterol levels moderately.[17]
There is insufficient evidence to determine the role of ingested capsaicin on several human disorders, including obesity, diabetes, cancer and cardiovascular diseases.[7]
Pepper spray and pests
Capsaicinoids are also an active ingredient in riot control and personal defense pepper spray agents.[18][19][20] When the spray comes in contact with skin, especially eyes or mucous membranes, it produces pain and breathing difficulty in the affected individual.
Capsaicin is also used to deter pests, specifically mammalian pests. Targets of capsaicin repellants include voles, deer, rabbits, squirrels, bears, insects, and attacking dogs.[21] Ground or crushed dried chili pods may be used in birdseed to deter rodents,[22] taking advantage of the insensitivity of birds to capsaicin. The Elephant Pepper Development Trust claims that using chili peppers as a barrier crop can be a sustainable means for rural African farmers to deter elephants from eating their crops.[23]
An article published in the Journal of Environmental Science and Health Part B in 2006 states that "Although hot chili pepper extract is commonly used as a component of household and garden insect-repellent formulas, it is not clear that the capsaicinoid elements of the extract are responsible for its repellency."[24]
The first pesticide product using solely capsaicin as the active ingredient was registered with the U.S. Department of Agriculture in 1962.[21]
Horse sports
Capsaicin is a banned substance in equestrian sports because of its hypersensitizing and pain-relieving properties. At the show jumping events of the 2008 Summer Olympics, four horses tested positive for capsaicin, which resulted in disqualification.[25]
Toxicity
Acute health effects
Capsaicin is a strong irritant requiring proper protective goggles, respirators, and proper hazardous material-handling procedures. Capsaicin takes effect upon skin contact (irritant, sensitizer), eye contact (irritant), ingestion, and inhalation (lung irritant, lung sensitizer). LD50 in mice is 47.2 mg/kg.[26][27]
Painful exposures to capsaicin-containing peppers are among the most common plant-related exposures presented to poison centers.[28] They cause burning or stinging pain to the skin and, if ingested in large amounts by adults or small amounts by children, can produce nausea, vomiting, abdominal pain, and burning diarrhea. Eye exposure produces intense tearing, pain, conjunctivitis, and blepharospasm.[29]
Treatment after exposure
The primary treatment is removal from exposure. Contaminated clothing should be removed and placed in airtight bags before incineration to prevent secondary exposure.
For external exposure, bathing the mucous membrane surfaces that have contacted capsaicin with oily compounds such as vegetable oil, paraffin oil, petroleum jelly (Vaseline), creams, or polyethylene glycol is the most effective way to attenuate the associated discomfort; since oil and capsaicin are both hydrophobic hydrocarbons, the capsaicin that has not already been absorbed into tissues will be picked up into solution and easily removed. Capsaicin can also be washed off the skin using soap, shampoo, or other detergents. Plain water is ineffective at removing capsaicin.[26] Capsaicin is soluble in alcohol, which can be used to clean contaminated items.[26]
When capsaicin is ingested, cold milk is an effective way to relieve the burning sensation (due to caseins, a protein found in milk, having a detergent effect on capsaicin[30]), and sugar solution (10%) at 20 °C (68 °F) is almost as effective.[31] The burning sensation will slowly fade away over several hours if no actions are taken.
Capsaicin-induced asthma might be treated with oral antihistamines or corticosteroids.[29]
Weight loss and regain
As of 2007, there was no evidence showing that weight loss is directly correlated with ingesting capsaicin. Well-designed clinical research had not been performed because the pungency of capsaicin in prescribed doses under research prevented subjects from complying in the study.[32] A 2014 meta-analysis of further trials found weak evidence that consuming capsaicin before a meal might slightly reduce the amount of food consumed, and might drive food preference toward carbohydrates.[33]
Peptic ulcer
One 2006 review concluded that capsaicin may relieve symptoms of a peptic ulcer rather than being a cause of it.[34]
Mechanism of action
The burning and painful sensations associated with capsaicin result from its chemical interaction with sensory neurons. Capsaicin, as a member of the vanilloid family, binds to a receptor called the vanilloid receptor subtype 1 (TRPV1).[35] First cloned in 1997, TRPV1 is an ion channel-type receptor.[36] TRPV1, which can also be stimulated with heat, protons and physical abrasion, permits cations to pass through the cell membrane when activated. The resulting depolarization of the neuron stimulates it to signal the brain. By binding to the TRPV1 receptor, the capsaicin molecule produces similar sensations to those of excessive heat or abrasive damage, explaining why the spiciness of capsaicin is described as a burning sensation.
Early research showed capsaicin to evoke a long-onset current in comparison to other chemical agonists, suggesting the involvement of a significant rate-limiting factor.[37] Subsequent to this, the TRPV1 ion channel has been shown to be a member of the superfamily of TRP ion channels, and as such is now referred to as TRPV1. There are a number of different TRP ion channels that have been shown to be sensitive to different ranges of temperature and probably are responsible for the human range of temperature sensation. Thus, capsaicin does not actually cause a chemical burn, or indeed any direct tissue damage at all, when chili peppers are the source of exposure. The inflammation resulting from exposure to capsaicin is believed to be the result of the body's reaction to nerve excitement. For example, the mode of action of capsaicin in inducing bronchoconstriction is thought to involve stimulation of C fibers[38] culminating in the release of neuropeptides. In essence, the body inflames tissues as if it has undergone a burn or abrasion and the resulting inflammation can cause tissue damage in cases of extreme exposure, as is the case for many substances that cause the body to trigger an inflammatory response.
Capsaicin was instrumental in the 2021 Nobel Prize in Physiology or Medicine, as it had led to the discovery of receptors for temperature and touch.[39][40]
Natural function
Capsaicin is present in large quantities in the placental tissue (which holds the seeds), the internal membranes and, to a lesser extent, the other fleshy parts of the fruits of plants in the genus Capsicum. The seeds themselves do not produce any capsaicin, although the highest concentration of capsaicin can be found in the white pith of the inner wall, where the seeds are attached.[41]
The seeds of Capsicum plants are dispersed predominantly by birds. In birds, the TRPV1 channel does not respond to capsaicin or related chemicals (avian vs. mammalian TRPV1 show functional diversity and selective sensitivity). This is advantageous to the plant, as chili pepper seeds consumed by birds pass through the digestive tract and can germinate later, whereas mammals have molar teeth which destroy such seeds and prevent them from germinating. Thus, natural selection may have led to increasing capsaicin production because it makes the plant less likely to be eaten by animals that do not help it disperse.[42] There is also evidence that capsaicin may have evolved as an anti-fungal agent.[43] The fungal pathogen Fusarium, which is known to infect wild chilies and thereby reduce seed viability, is deterred by capsaicin, which thus limits this form of predispersal seed mortality.
The vanillotoxin-containing venom of a certain tarantula species (Psalmopoeus cambridgei,[44] also known as the Trinidad chevron tarantula) activates the same pathway of pain as is activated by capsaicin, an example of a shared pathway in both plant and animal anti-mammalian defense.[44]
History
The compound was first extracted in impure form in 1816 by Christian Friedrich Bucholz (1770–1818).[45][lower-alpha 1] He called it "capsicin", after the genus Capsicum from which it was extracted. John Clough Thresh (1850–1932), who had isolated capsaicin in almost pure form,[46][47] gave it the name "capsaicin" in 1876.[48] Karl Micko isolated capsaicin in its pure form in 1898.[49][50] Capsaicin's chemical composition was first determined in 1919 by E. K. Nelson, who also partially elucidated capsaicin's chemical structure.[51] Capsaicin was first synthesized in 1930 by Ernst Spath and Stephen F. Darling.[52] In 1961, similar substances were isolated from chili peppers by the Japanese chemists S. Kosuge and Y. Inagaki, who named them capsaicinoids.[53][54]
In 1873 German pharmacologist Rudolf Buchheim[55] (1820–1879) and in 1878 the Hungarian doctor Endre Hőgyes[56] stated that "capsicol" (partially purified capsaicin[57]) caused the burning feeling when in contact with mucous membranes and increased secretion of gastric acid.
Capsaicinoids
The most commonly occurring capsaicinoids are capsaicin (69%), dihydrocapsaicin (22%), nordihydrocapsaicin (7%), homocapsaicin (1%), and homodihydrocapsaicin (1%).[58]
Capsaicin and dihydrocapsaicin (both 16.0 million SHU) are the most pungent capsaicinoids. Nordihydrocapsaicin (9.1 million SHU), homocapsaicin and homodihydrocapsaicin (both 8.6 million SHU) are about half as hot.[3]
There are six natural capsaicinoids (table below). Although vanillylamide of n-nonanoic acid (Nonivamide, VNA, also PAVA) is produced synthetically for most applications, it does occur naturally in Capsicum species.[59]
| Capsaicinoid name | Abbrev. | Typical relative amount |
Scoville heat units |
Chemical structure |
|---|---|---|---|---|
| Capsaicin | C | 69% | 16,000,000 |  |
| Dihydrocapsaicin | DHC | 22% | 16,000,000 | 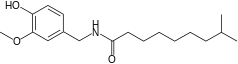 |
| Nordihydrocapsaicin | NDHC | 7% | 9,100,000 | 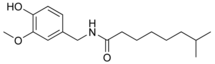 |
| Homocapsaicin | HC | 1% | 8,600,000 | 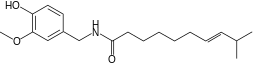 |
| Homodihydrocapsaicin | HDHC | 1% | 8,600,000 | 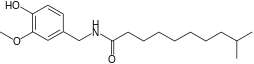 |
| Nonivamide | PAVA | 9,200,000 | 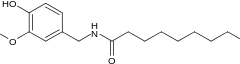 |
Biosynthesis




History
The general biosynthetic pathway of capsaicin and other capsaicinoids was elucidated in the 1960s by Bennett and Kirby, and Leete and Louden. Radiolabeling studies identified phenylalanine and valine as the precursors to capsaicin.[60][61] Enzymes of the phenylpropanoid pathway, phenylalanine ammonia lyase (PAL), cinnamate 4-hydroxylase (C4H), caffeic acid O-methyltransferase (COMT) and their function in capsaicinoid biosynthesis were identified later by Fujiwake et al.,[62][63] and Sukrasno and Yeoman.[64] Suzuki et al. are responsible for identifying leucine as another precursor to the branched-chain fatty acid pathway.[65] It was discovered in 1999 that pungency of chili peppers is related to higher transcription levels of key enzymes of the phenylpropanoid pathway, phenylalanine ammonia lyase, cinnamate 4-hydroxylase, caffeic acid O-methyltransferase. Similar studies showed high transcription levels in the placenta of chili peppers with high pungency of genes responsible for branched-chain fatty acid pathway.[66]
Biosynthetic pathway
Plants exclusively of the genus Capsicum produce capsaicinoids, which are alkaloids.[67] Capsaicin is believed to be synthesized in the interlocular septum of chili peppers and depends on the gene AT3, which resides at the pun1 locus, and which encodes a putative acyltransferase.[68]
Biosynthesis of the capsaicinoids occurs in the glands of the pepper fruit where capsaicin synthase condenses vanillylamine from the phenylpropanoid pathway with an acyl-CoA moiety produced by the branched-chain fatty acid pathway.[69][70][71][72]
Capsaicin is the most abundant capsaicinoid found in the genus Capsicum, but at least ten other capsaicinoid variants exist.[73] Phenylalanine supplies the precursor to the phenylpropanoid pathway while leucine or valine provide the precursor for the branched-chain fatty acid pathway.[69][70] To produce capsaicin, 8-methyl-6-nonenoyl-CoA is produced by the branched-chain fatty acid pathway and condensed with vanillamine. Other capsaicinoids are produced by the condensation of vanillamine with various acyl-CoA products from the branched-chain fatty acid pathway, which is capable of producing a variety of acyl-CoA moieties of different chain length and degrees of unsaturation.[74] All condensation reactions between the products of the phenylpropanoid and branched-chain fatty acid pathway are mediated by capsaicin synthase to produce the final capsacinoid product.[69][70]
See also
- Allicin, the active piquant flavor chemical in uncooked garlic, and to a lesser extent onions (see those articles for discussion of other chemicals in them relating to pungency, and eye irritation)
- Capsazepine, capsaicin antagonist
- Iodoresiniferatoxin, an ultrapotent capsaicin antagonist derived from Resiniferatoxin
- Naga Viper pepper, Bhut Jolokia Pepper, Carolina Reaper, Trinidad Moruga Scorpion; some of the world's most capsaicin-rich fruits
References
- ↑ "Capsaicin". ChemSpider, Royal Society of Chemistry, Cambridge, UK. 2018. Archived from the original on 12 June 2018. Retrieved 9 June 2018.
- 1 2 3 4 "Capsaicin, Experimental Properties". PubChem, US National Library of Medicine. 2 June 2018. Archived from the original on 12 June 2018. Retrieved 9 June 2018.
- 1 2 Govindarajan, V. S.; Sathyanarayana, M. N. (1991). "Capsicum—production, technology, chemistry, and quality. Part V. Impact on physiology, pharmacology, nutrition, and metabolism; structure, pungency, pain, and desensitization sequences". Critical Reviews in Food Science and Nutrition. 29 (6): 435–74. doi:10.1080/10408399109527536. PMID 2039598.
- ↑ What Made Chili Peppers So Spicy? Archived 12 May 2021 at the Wayback Machine Talk of the Nation, 15 August 2008.
- 1 2 3 4 Gorman J (20 September 2010). "A Perk of Our Evolution: Pleasure in Pain of Chilies". New York Times. Archived from the original on 2 April 2015. Retrieved 16 March 2015.
- 1 2 Rollyson WD, et al. (2014). "Bioavailability of capsaicin and its implications for drug delivery". J Control Release. 196: 96–105. doi:10.1016/j.jconrel.2014.09.027. PMC 4267963. PMID 25307998.
- 1 2 3 4 5 Fattori, V; Hohmann, M. S.; Rossaneis, A. C.; Pinho Ribeiro, F. A.; Verri, W. A. (2016). "Capsaicin: Current Understanding of Its Mechanisms and Therapy of Pain and Other Pre-Clinical and Clinical Uses". Molecules. 21 (7): 844. doi:10.3390/molecules21070844. PMC 6273101. PMID 27367653.
- ↑ Ton, Joey (10 December 2018). "#225 Topical Capsaicin for Neuropathic and Osteoarthritis Pain: Maybe not so hot?". CFPCLearn. Archived from the original on 30 January 2023. Retrieved 16 June 2023.
- ↑ "FDA Approves New Drug Treatment for Long-Term Pain Relief after Shingles Attacks" (Press release). U.S. Food and Drug Administration. 17 November 2009. Archived from the original on 23 November 2015. Retrieved 5 January 2016.
- ↑ "Drug Approval Package: Qutenza (capsaicin) NDA #022395". U.S. Food and Drug Administration (FDA). 29 June 2010. Archived from the original on 28 November 2020. Retrieved 19 August 2020.
- "Application Number: 22-395: Summary Review" (PDF). FDA Center for Drug Evaluation and Research. November 13, 2009. Archived (PDF) from the original on 17 October 2022. Retrieved 9 December 2022.
- ↑ "Qutenza EPAR". European Medicines Agency (EMA). Archived from the original on 4 September 2020. Retrieved 19 August 2020.
- ↑ Hitt E (9 March 2012). "FDA Turns Down Capsaicin Patch for Painful Neuropathy in HIV". Medscape Medical News, WebMD. Archived from the original on 1 June 2012. Retrieved 5 January 2016.
- ↑ Derry, S.; Rice, A. S.; Cole, P.; Tan, T.; Moore, R. A. (2017). "Topical capsaicin (high concentration) for chronic neuropathic pain in adults" (PDF). The Cochrane Database of Systematic Reviews. 1 (7): CD007393. doi:10.1002/14651858.CD007393.pub4. hdl:10044/1/49554. PMC 6464756. PMID 28085183. Archived from the original (PDF) on 15 February 2021. Retrieved 27 September 2018.
- ↑ Glinski W, Glinska-Ferenz M, Pierozynska-Dubowska M (1991). "Neurogenic inflammation induced by capsaicin in patients with psoriasis". Acta Dermato-Venereologica. 71 (1): 51–4. PMID 1711752.
- ↑ Ellis CN, Berberian B, Sulica VI, Dodd WA, Jarratt MT, Katz HI, Prawer S, Krueger G, Rex IH Jr, Wolf JE (1993). "A double-blind evaluation of topical capsaicin in pruritic psoriasis". J. Am. Acad. Dermatol. 29 (3): 438–42. doi:10.1016/0190-9622(93)70208-B. PMID 7688774.
- ↑ Gooding SM, Canter PH, Coelho HF, Boddy K, Ernst E (2010). "Systematic review of topical capsaicin in the treatment of pruritus". Int J Dermatol. 49 (8): 858–65. doi:10.1111/j.1365-4632.2010.04537.x. PMID 21128913. S2CID 24484878.
- ↑ Kelava L, Nemeth D, Hegyi P, Keringer P, Kovacs DK, Balasko M, Solymar M, Pakai E, Rumbus Z, Garami A (April 2021). "Dietary supplementation of transient receptor potential vanilloid-1 channel agonists reduces serum total cholesterol level: a meta-analysis of controlled human trials". Critical Reviews in Food Science and Nutrition. 62 (25): 7025–7035. doi:10.1080/10408398.2021.1910138. PMID 33840333.
- ↑ Blum, Deborah (21 November 2011). "About Pepper Spray". Scientific-American Guest Blog (Web). London, UK: Macmillan Publishers, Ltd. Archived from the original on 17 February 2016. Retrieved 17 February 2016.
- ↑ "Capsaicin". Toxicology Data Network (Database). Bethesda, MD: National Institutes of Health (published 30 June 2006). 15 March 1983. Archived from the original on 13 May 2016. Retrieved 17 February 2016.
- ↑ Smith, C. Gregory; Stopford, Woodhall (1999). "Health Hazards of Pepper Spray". North Carolina Medical Journal (Web). Morrisville, NC: North Carolina Institute of Medicine. 60 (5): 268–74. PMID 10495655. Archived from the original on 17 August 2000.
- 1 2 "R.E.D. Facts for Capsaicin" (PDF). United States Environmental Protection Agency. Archived from the original (PDF) on 24 October 2012. Retrieved 13 November 2012.
- ↑ Jensen, P. G.; Curtis, P. D.; Dunn, J. A.; Austic, R. E.; Richmond, M. E. (2003). "Field evaluation of capsaicin as a rodent aversion agent for poultry feed". Pest Management Science. 59 (9): 1007–1015. doi:10.1002/ps.705. PMID 12974352.
- ↑ "Elephant Pepper: Human Elephant Conflict and Chilli Pepper". www.elephantpepper.org. Archived from the original on 31 May 2019. Retrieved 31 May 2019.
- ↑ Antonious GF, Meyer JE, Snyder JC (2006). "Toxicity and repellency of hot pepper extracts to spider mite, Tetranychus urticae Koch". J Environ Sci Health B. 41 (8): 1383–91. doi:10.1080/0360123060096419. PMID 17090499. S2CID 19121573.
- ↑ "Olympic horses fail drugs tests". BBC News. 21 August 2008. Archived from the original on 12 May 2009. Retrieved 1 April 2010.
- 1 2 3 "Capsaicin Material Safety Data Sheet". sciencelab.com. 2007. Archived from the original (PDF) on 29 September 2007. Retrieved 13 July 2007.
- ↑ Johnson, Wilbur (2007). "Final report on the safety assessment of Capsicum annuum extract, Capsicum annuum fruit extract, Capsicum annuum resin, Capsicum annuum fruit powder, Capsicum frutescens fruit, Capsicum frutescens fruit extract, Capsicum frutescens resin, and capsaicin". Int. J. Toxicol. 26 Suppl 1: 3–106. doi:10.1080/10915810601163939. PMID 17365137. S2CID 208154058.
- ↑ Krenzelok, E. P.; Jacobsen, T. D. (1997-08-01). "Plant exposures: a national profile of the most common plant genera". Veterinary and Human Toxicology. 39 (4): 248–249. ISSN 0145-6296. PMID 9251180.
- 1 2 Goldfrank, L R., ed. (2007-03-23). Goldfrank's Toxicologic Emergencies. New York, New York: McGraw-Hill. p. 1167. ISBN 978-0-07-144310-4.
- ↑ "General Chemistry Online: Fire and Spice". antoine.frostburg.edu. Archived from the original on 16 October 2022. Retrieved 9 December 2022.
- ↑ Wu Nasrawia, Christina; Marie Pangborn, Rose (April 1990). "Temporal effectiveness of mouth-rinsing on capsaicin mouth-burn". Physiol. Behav. 47 (4): 617–23. doi:10.1016/0031-9384(90)90067-E. PMID 2385629. S2CID 40829476.
- ↑ Diepvens K, Westerterp KR, Westerterp-Plantenga MS (2007). "Obesity and thermogenesis related to the consumption of caffeine, ephedrine, capsaicin, and green tea". American Journal of Physiology. Regulatory, Integrative and Comparative Physiology. 292 (1): R77–85. doi:10.1152/ajpregu.00832.2005. PMID 16840650. S2CID 7529851. Archived from the original on 26 December 2022. Retrieved 9 December 2022.
- ↑ Whiting, S; Derbyshire, EJ; Tiwari, B (February 2014). "Could capsaicinoids help to support weight management? A systematic review and meta-analysis of energy intake data". Appetite. 73: 183–8. doi:10.1016/j.appet.2013.11.005. PMID 24246368. S2CID 30252935.
- ↑ Satyanarayana MN (2006). "Capsaicin and gastric ulcers". Critical Reviews in Food Science and Nutrition. 46 (4): 275–328. doi:10.1080/1040-830491379236. PMID 16621751. S2CID 40023195.
- ↑ Story GM, Crus-Orengo L (July–August 2007). "Feel the burn". American Scientist. 95 (4): 326–333. doi:10.1511/2007.66.326.
- ↑ Caterina, MJ; Schumacher, MA; Tominaga, M; Rosen, TA; Levine, JD; Julius, D (23 October 1997). "The capsaicin receptor: a heat-activated ion channel in the pain pathway". Nature. 389 (6653): 816–24. Bibcode:1997Natur.389..816C. doi:10.1038/39807. PMID 9349813. S2CID 7970319.
- ↑ Geppetti, Pierangelo & Holzer, Peter (1996). Neurogenic Inflammation. CRC Press, 1996.
- ↑ Fuller, R. W., Dixon, C. M. S. & Barnes, P. J. (1985). "Bronchoconstrictor response to inhaled capsaicin in humans" J. Appl. Physiol 58, 1080–1084. PubMed, CAS, Web of Science® Times Cited: 174
- ↑ "The Nobel Prize in Physiology or Medicine 2021". NobelPrize.org. Archived from the original on 4 October 2021. Retrieved 9 December 2022.
- ↑ Santora, Marc; Engelbrecht, Cora (4 October 2021). "Nobel Prize Awarded to Scientists for Research About Temperature and Touch". The New York Times. Archived from the original on 3 December 2022. Retrieved 9 December 2022.
- ↑ New Mexico State University – College of Agriculture and Home Economics (2005). "Chile Information – Frequently Asked Questions". Archived from the original on 4 May 2007. Retrieved 17 May 2007.
- ↑ Tewksbury, J. J.; Nabhan, G. P. (2001). "Seed dispersal. Directed deterrence by capsaicin in chilies". Nature. 412 (6845): 403–404. doi:10.1038/35086653. PMID 11473305. S2CID 4389051.
- ↑ Joshua J. Tewksbury; Karen M. Reagan; Noelle J. Machnicki; Tomás A. Carlo; David C. Haak; Alejandra Lorena Calderón Peñaloza; Douglas J. Levey (19 August 2008), "Evolutionary ecology of pungency in wild chilies", Proceedings of the National Academy of Sciences, 105 (33): 11808–11811, Bibcode:2008PNAS..10511808T, doi:10.1073/pnas.0802691105, PMC 2575311, PMID 18695236
- 1 2 Siemens J, Zhou S, Piskorowski R, et al. (November 2006). "Spider toxins activate the capsaicin receptor to produce inflammatory pain". Nature. 444 (7116): 208–12. Bibcode:2006Natur.444..208S. doi:10.1038/nature05285. PMID 17093448. S2CID 4387600.
- ↑ Bucholz, C. F. (1816). "Chemische Untersuchung der trockenen reifen spanischen Pfeffers" [Chemical investigation of dry, ripe Spanish peppers]. Almanach oder Taschenbuch für Scheidekünstler und Apotheker [Almanac or Pocketbook for Analysts and Apothecaries]. Vol. 37. Weimar. pp. 1–30. [Note: Christian Friedrich Bucholz's surname has been variously spelled as "Bucholz", "Bucholtz", or "Buchholz".]
- ↑ In a series of articles, J. C. Thresh obtained capsaicin in almost pure form:
- J. C. Thresh (1876) "Isolation of capsaicin," The Pharmaceutical Journal and Transactions, 3rd series, vol. 6, pages 941–947;
- J. C. Thresh (8 July 1876) "Capsaicin, the active principle in Capsicum fruits," The Pharmaceutical Journal and Transactions, 3rd series, vol. 7, no. 315, pages 21 ff. [Note: This article is summarized in: "Capsaicin, the active principle in Capsicum fruits," The Analyst, vol. 1, no. 8, pages 148–149 Archived 20 September 2020 at the Wayback Machine, (1876).]. In The Pharmaceutical Journal and Transactions, volume 7, see also pages 259ff and 473 ff and in vol. 8, see pages 187ff;
- Year Book of Pharmacy… (1876), pages 250 and 543;
- J. C. Thresh (1877) "Note on Capsaicin," Year Book of Pharmacy…, pages 24–25;
- J. C. Thresh (1877) "Report on the active principle of Cayenne pepper," Year Book of Pharmacy..., pages 485–488.
- ↑ Obituary notice of J. C. Thresh: "John Clough Thresh, M.D., D. Sc., and D.P.H". The British Medical Journal. 1 (3726): 1057–1058. 1932. doi:10.1136/bmj.1.3726.1057-c. PMC 2521090. PMID 20776886.
- ↑ J King, H Wickes Felter, J Uri Lloyd (1905) A King's American Dispensatory. Eclectic Medical Publications (ISBN 1888483024)
- ↑ Micko K (1898). "Zur Kenntniss des Capsaïcins" [On our knowledge of capsaicin]. Zeitschrift für Untersuchung der Nahrungs- und Genussmittel (Journal for the Investigation of Necessities and Luxuries) (in Deutsch). 1 (12): 818–829. doi:10.1007/bf02529190.
- ↑ Karl Micko (1899). "Über den wirksamen Bestandtheil des Cayennespfeffers" [On the active component of Cayenne pepper]. Zeitschrift für Untersuchung der Nahrungs- und Genussmittel (in Deutsch). 2 (5): 411–412. doi:10.1007/bf02529197.
- ↑ Nelson EK (1919). "The constitution of capsaicin, the pungent principle of capsicum". J. Am. Chem. Soc. 41 (7): 1115–1121. doi:10.1021/ja02228a011. Archived from the original on 26 December 2022. Retrieved 9 December 2022.
- ↑ Späth E, Darling SF (1930). "Synthese des Capsaicins". Chem. Ber. 63B (3): 737–743. doi:10.1002/cber.19300630331.
- ↑ S Kosuge, Y Inagaki, H Okumura (1961). Studies on the pungent principles of red pepper. Part VIII. On the chemical constitutions of the pungent principles. Nippon Nogei Kagaku Kaishi (J. Agric. Chem. Soc.), 35, 923–927; (en) Chem. Abstr. 1964; 60, 9827g.
- ↑ (ja) S Kosuge, Y Inagaki (1962) Studies on the pungent principles of red pepper. Part XI. Determination and contents of the two pungent principles. Nippon Nogei Kagaku Kaishi J. Agric. Chem. Soc., 36, pp. 251
- ↑ Rudolf Buchheim (1873) "Über die 'scharfen' Stoffe" (On the "hot" substance), Archiv der Heilkunde (Archive of Medicine), vol. 14, pages 1ff. See also: R. Buchheim (1872) "Fructus Capsici," Vierteljahresschrift für praktische Pharmazie (Quarterly Journal for Practical Pharmacy), vol. 4, pages 507ff.; reprinted (in English) in: Proceedings of the American Pharmaceutical Association, vol. 22, pages 106ff (1873).
- ↑ Endre Hőgyes, "Adatok a paprika (Capsicum annuum) élettani hatásához" [Data on the physiological effects of the pepper (Capsicum annuum)], Orvos-természettudumányi társulatot Értesítője [Bulletin of the Medical Science Association] (1877); reprinted in: Orvosi Hetilap [Medical Journal] (1878), 10 pages. Published in German as: "Beitrage zur physiologischen Wirkung der Bestandtheile des Capiscum annuum (Spanischer Pfeffer)" [Contributions on the physiological effects of components of Capsicum annuum (Spanish pepper)], Archiv für Experimentelle Pathologie und Pharmakologie, vol. 9, pages 117–130 (1878). See springerlink.com Archived 26 December 2022 at the Wayback Machine
- ↑ F.A. Flückiger, Pharmakognosie des Pflanzenreiches ( Berlin, Germany: Gaertner's Verlagsbuchhandlung, 1891).
- ↑ Bennett DJ, Kirby GW (1968). "Constitution and biosynthesis of capsaicin". J. Chem. Soc. C: 442. doi:10.1039/j39680000442.
- ↑ Howard L. Constant, Geoffrey A. Cordell and Dennis P. West (1996). "Nonivamide, a Constituent of Capsicum oleoresin". J. Nat. Prod. 59 (4): 425–426. doi:10.1021/np9600816.
- ↑ Bennett DJ, Kirby GW (1968) Constitution and biosynthesis of capsaicin. J Chem Soc C 4:442–446
- ↑ Leete E, Louden MC (1968). "Biosynthesis of capsaicin and dihydrocapsaicin in Capsicum frutescens". J Am Chem Soc. 90 (24): 6837–6841. doi:10.1021/ja01026a049. PMID 5687710.
- ↑ Fujiwake H, Suzuki T, Iwai K (1982a) Intracellular distribution of enzymes and intermediates involved in biosynthesis of capsaicin and its analogues in Capsicum fruits. Agric Biol Chem 46:2685–2689
- ↑ Fujiwake H, Suzuki T, Iwai K (1982b) Capsaicinoid formation in the protoplast from placenta of Capsicum fruits. Agric Biol Chem 46:2591–2592
- ↑ Sukrasno N, Yeoman MM (1993). "Phenylpropanoid metabolism during growth and development of Capsicum frutescens fruits". Phytochemistry. 32 (4): 839–844. doi:10.1016/0031-9422(93)85217-f.
- ↑ Suzuki, T; Kawada, T; Iwai, K (1981). "Formation and metabolism of pungent principle of Capsicum fruits. 9. Biosynthesis of acyl moieties of capsaicin and its analogs from valine and leucine in Capsicum fruits". Plant Cell Physiol. 22: 23–32.
- ↑ Curry, J; Aluru, M; Mendoza, M; Nevarez, J; Melendrez, M; O'Connell, MA (1999). "Transcripts for possible capsaicinoid biosynthetic genes are differentially accumulated in pungent and non-pungent Capsicum spp". Plant Sci. 148: 47–57. doi:10.1016/s0168-9452(99)00118-1. S2CID 86735106.
- ↑ Nelson EK, Dawson LE (1923). "Constitution of capsaicin, the pungent principle of Capsicum. III". J Am Chem Soc. 45 (9): 2179–2181. doi:10.1021/ja01662a023.
- ↑ Stewart C, Kang BC, Liu K, et al. (June 2005). "The Pun1 gene for pungency in pepper encodes a putative acyltransferase". Plant J. 42 (5): 675–88. doi:10.1111/j.1365-313X.2005.02410.x. PMID 15918882.
- 1 2 3 Leete E, Louden MC (1968). "Biosynthesis of capsaicin and dihydrocapsaicin in Capsicum frutescens". J. Am. Chem. Soc. 90 (24): 6837–6841. doi:10.1021/ja01026a049. PMID 5687710.
- 1 2 3 Bennett DJ, Kirby GW (1968). "Constitution and biosynthesis of capsaicin". J. Chem. Soc. C. 1968: 442–446. doi:10.1039/j39680000442.
- ↑ Fujiwake H.; Suzuki T.; Oka S.; Iwai K. (1980). "Enzymatic formation of capsaicinoid from vanillylamine and iso-type fatty acids by cell-free extracts of Capsicum annuum var. annuum cv. Karayatsubusa". Agricultural and Biological Chemistry. 44 (12): 2907–2912. doi:10.1271/bbb1961.44.2907.
- ↑ I. Guzman, P.W. Bosland, and M.A. O'Connell, "Chapter 8: Heat, Color, and Flavor Compounds in Capsicum Fruit" in David R. Gang, ed., Recent Advances in Phytochemistry 41: The Biological Activity of Phytochemicals (New York, New York: Springer, 2011), pages 117–118.
- ↑ Kozukue N, Han JS, Kozukue E, Lee SJ, Kim JA, Lee KR, Levin CE, Friedman M (2005). "Analysis of eight capsaicinoids in peppers and pepper-containing foods by high-performance liquid chromatography and liquid chromatography-mass spectrometry". J Agric Food Chem. 53 (23): 9172–9181. doi:10.1021/jf050469j. PMID 16277419.
- ↑ Thiele R, Mueller-Seitz E, Petz M (2008). "Chili pepper fruits: presumed precursors of fatty acids characteristic for capsaicinoids". J Agric Food Chem. 56 (11): 4219–4224. doi:10.1021/jf073420h. PMID 18489121.
Notes
- ↑ History of early research on capsaicin:
- Felter, Harvey W.; Lloyd, John U. (1898). King's American Dispensatory. Vol. 1. Cincinnati, Ohio: Ohio Valley Co. p. 435. Archived from the original on 13 May 2008. Retrieved 9 December 2022.
- Du Mez, Andrew G. (1917). A century of the United States pharmocopoeia 1820–1920. I. The galenical oleoresins (PhD). University of Wisconsin. pp. 111–132.
- The results of Bucholz's and Braconnot's analyses of Capsicum annuum appear in: Pereira, Jonathan (1854). The Elements of Materia Medica and Therapeutics. Vol. 2 (3rd US ed.). Philadelphia, Pennsylvania: Blanchard and Lea. p. 506. Archived from the original on 26 December 2022. Retrieved 9 December 2022.
- Biographical information about Christian Friedrich Bucholz is available in: Rose, Hugh J.; Rose, Henry J.; Wright, Thomas, eds. (1857). A New General Biographical Dictionary. Vol. 5. London, England. p. 186. Archived from the original on 26 December 2022. Retrieved 9 December 2022.
- Biographical information about C. F. Bucholz is also available (in German) online at: Allgemeine Deutsche Biographie.
- Some other early investigators who also extracted the active component of peppers:
- Benjamin Maurach (1816) "Pharmaceutisch-chemische Untersuchung des spanischen Pfeffers" (Pharmaceutical-chemical investigation of Spanish peppers), Berlinisches Jahrbuch für die Pharmacie, vol. 17, pages 63–73. Abstracts of Maurach's paper appear in: (i) Repertorium für die Pharmacie, vol. 6, page 117-119 (1819); (ii) Allgemeine Literatur-Zeitung, vol. 4, no. 18, page 146 (Feb. 1821); (iii) "Spanischer oder indischer Pfeffer", System der Materia medica ..., vol. 6, pages 381–386 (1821) (this reference also contains an abstract of Bucholz's analysis of peppers).
- French chemist Henri Braconnot (1817) "Examen chemique du Piment, de son principe âcre, et de celui des plantes de la famille des renonculacées" (Chemical investigation of the chili pepper, of its pungent principle [constituent, component], and of that of plants of the family Ranunculus), Annales de Chemie et de Physique, vol. 6, pages 122- 131.
- Danish geologist Johann Georg Forchhammer in: Hans C. Oersted (1820) "Sur la découverte de deux nouveaux alcalis végétaux" (On the discovery of two new plant alkalis), Journal de physique, de chemie, d'histoire naturelle et des arts, vol. 90, pages 173–174.
- German apothecary Ernst Witting (1822) "Considerations sur les bases vegetales en general, sous le point de vue pharmaceutique et descriptif de deux substances, la capsicine et la nicotianine" (Thoughts on the plant bases in general from a pharmaceutical viewpoint, and description of two substances, capsicin and nicotine), Beiträge für die pharmaceutische und analytische Chemie, vol. 3, pages 43ff.
Further reading
- Abdel-Salam, Omar M. E., ed.: Capsaicin as a Therapeutic Molecule. Springer, 2014. ISBN 978-3-0348-0827-9 (print); ISBN 978-3-0348-0828-6 (eBook).
External links
| Look up capsaicin in Wiktionary, the free dictionary. |
- Capsaicin Technical Fact Sheet – National Pesticide Information Center Archived 1 July 2015 at the Wayback Machine
- Fire and Spice: The molecular basis for flavor Archived 5 July 2011 at the Wayback Machine

