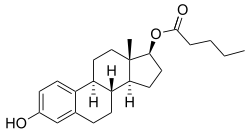
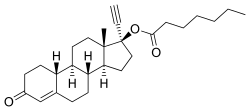
Estradiol valerate/norethisterone enantate
 | |
 | |
| Combination of | |
|---|---|
| Estradiol valerate | Estrogen |
| Norethisterone enantate | Progestogen |
| Clinical data | |
| Trade names | Mesigyna, others |
| Other names | EV/NETE; HRP-102 |
| Routes of administration | Intramuscular injection |
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| UNII | |
Estradiol valerate/norethisterone enantate (EV/NETE), sold under the brand name Mesigyna among others, is a form of combined injectable birth control which is used to prevent pregnancy in women.[1][2][3][4] It contains estradiol valerate (EV), an estrogen, and norethisterone enantate (NETE), a progestin.[1][2][3] The medication is given once a month by injection into muscle.[1][2][3]
EV/NETE is approved for use in at least 36 countries, and is the most widely used combined injectable contraceptive.[5][3] It is available widely throughout Latin America, in a few Asian and African countries, and in Turkey.[6][7][8]
Medical uses
EV/NETE is used as a combined injectable contraceptive to prevent pregnancy in women.[1][2][3] It is given by intramuscular injection once a month.[1][2][3]
Available forms
EV/NETE is available in the form of an oil solution containing 5 mg estradiol valerate (EV) and 50 mg norethisterone enantate (NETE).[5][1][2][3]
Pharmacology
Pharmacokinetics
Peak levels of estradiol after an intramuscular injection of EV/NETE (5 mg/50 mg) are reached within 2 days and range from 232 to 428 pg/mL.[4][1]
History
EV/NETE, along with estradiol cypionate/medroxyprogesterone acetate (EC/MPA; code name HRP-112), was developed by the World Health Organization.[5][9] Both EV/NETE and EC/MPA became available in 1993.[10]
Society and culture
Generic names
EV/NETE is also known by its former developmental code name HRP-102.[11]
Brand names
EV/NETE has been marketed under a variety of brand names including Chinese Injectable No. 3, Effectimes, Ginediol, Mesigyna, Mesilar, Meslart, Mesocept, Mesygest, Nofertyl, Nofertyl Lafrancol, Noregyna, Norestrin, Norifam, Norigynon, Nostidyn, Sexseg, and Solouna.[6][7][8][12][3][13][9][1][14]
Availability
EV/NETE has been marketed in at least 36 countries, including Argentina, the Bahamas, Barbados, Bolivia, Brazil, Chile, Colombia, Costa Rica, the Dominican Republic, Ecuador, Egypt, El Salvador, Ghana, Grenada, Guatemala, Guyana, Haiti, Honduras, Jamaica, Kenya, Mexico, Nicaragua, Panama, Paraguay, Peru, St. Lucia, Turkey, Uruguay, Venezuela, and Zimbabwe.[6][7][8][12][3][13][9][1][14] At least 15 of the countries in which EV/NETE is registered are Caribbean states.[5] EV/NETE is the most widely used combined injectable contraceptive.[5][3]
See also
- Combined injectable birth control § Available forms
- Special Programme on Human Reproduction
- List of combined sex-hormonal preparations
References
- 1 2 3 4 5 6 7 8 9 Garza-Flores J (April 1994). "Pharmacokinetics of once-a-month injectable contraceptives". Contraception. 49 (4): 347–59. doi:10.1016/0010-7824(94)90032-9. PMID 8013219.
- 1 2 3 4 5 6 Sang GW (April 1994). "Pharmacodynamic effects of once-a-month combined injectable contraceptives". Contraception. 49 (4): 361–85. doi:10.1016/0010-7824(94)90033-7. PMID 8013220.
- 1 2 3 4 5 6 7 8 9 10 Bagade O, Pawar V, Patel R, Patel B, Awasarkar V, Diwate S (2014). "Increasing use of long-acting reversible contraception: safe, reliable, and cost-effective birth control" (PDF). World J Pharm Pharm Sci. 3 (10): 364–392. ISSN 2278-4357.
- 1 2 "Archived copy". www.farmaciasahumada.cl. Archived from the original on 18 November 2018. Retrieved 15 January 2022.
{{cite web}}: CS1 maint: archived copy as title (link) - 1 2 3 4 5 Hall PE (August 1998). "New once-a-month injectable contraceptives, with particular reference to Cyclofem/Cyclo-Provera". Int J Gynaecol Obstet. 62 Suppl 1: S43–56. doi:10.1016/S0020-7292(98)00090-3. PMID 9806239. S2CID 2826689.
- 1 2 3 "Norethisterone".
- 1 2 3 Sweetman, Sean C., ed. (2009). "Sex hormones and their modulators". Martindale: The Complete Drug Reference (36th ed.). London: Pharmaceutical Press. p. 2082. ISBN 978-0-85369-840-1.
- 1 2 3 "Micromedex Products: Please Login".
- 1 2 3 Newton JR, D'arcangues C, Hall PE (1994). "A review of "once-a-month" combined injectable contraceptives". J Obstet Gynaecol (Lahore). 4 Suppl 1: S1–34. doi:10.3109/01443619409027641. PMID 12290848.
- ↑ d'Arcangues C (1993). "Once-a-month injectable contraceptives". World Health Forum. 14 (4): 439–40. PMID 8185807.
- ↑ Koetsawang S (April 1994). "Once-a-month injectable contraceptives: efficacy and reasons for discontinuation". Contraception. 49 (4): 387–98. doi:10.1016/0010-7824(94)90034-5. PMID 8013221.
- 1 2 IARC Working Group on the Evaluation of Carcinogenic Risks to Humans; International Agency for Research on Cancer (1 January 1999). Hormonal Contraception and Post-menopausal Hormonal Therapy (PDF). IARC. p. 65. ISBN 978-92-832-1272-0.
- 1 2 Pramilla Senanayake; Malcolm Potts (14 April 2008). Atlas of Contraception, Second Edition. CRC Press. pp. 50–. ISBN 978-0-203-34732-4.
- 1 2 IARC Working Group on the Evaluation of Carcinogenic Risks to Humans; World Health Organization; International Agency for Research on Cancer (2007). Combined Estrogen-progestogen Contraceptives and Combined Estrogen-progestogen Menopausal Therapy. World Health Organization. pp. 431–. ISBN 978-92-832-1291-1.