Methylmethaqualone
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
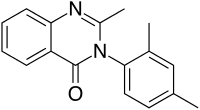
| Formula | C17H16N2O |
| Molar mass | 264.322 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
| (verify) | |
Methylmethaqualone (MMQ) is a quinazolinone and an analogue of methaqualone that has similar sedative and hypnotic properties to its parent compound (resulting from its agonist activity at the β subtype of the GABAA receptor), and is around the same potency. Methylmethaqualone differs from methaqualone by 4-methylation on the phenyl ring. It was made illegal in Germany in 1999 and listed by the DEA as a "drug of forensic interest" at about the same time, but little other information is available. It would appear that this compound was sold on the black market in Germany as a designer drug analogue of methaqualone.[1][2]
Animal studies of methylmethaqualone have shown it to produce convulsions at only slightly above the effective sedative dose,[3] and anecdotal reports from human users have confirmed that it can have a pro-convulsive effect, which has potential to make this compound particularly hazardous if taken in excessive doses.
References
- ↑ Klein RF, Hays PA (January–June 2003). "Detection and Analysis of Drugs of Forensic Interest, 1992 - 2001; A Literature Review" (PDF). Microgram Journal. DEA. 1 (1–2): 60. Archived from the original (PDF) on 2011-07-19.
- ↑ Angelos SA, Lankin DC, Meyers JA, Raney JK (March 1993). "The structural identification of a methyl analog of methaqualone via 2-dimensional NMR techniques". Journal of Forensic Sciences. 38 (2): 455–65. doi:10.1520/JFS13428J. PMID 8455002.
- ↑ Boltze KH, Dell HD, Lehwald H, Lorenz D, Rueberg-Schweer M (August 1963). "[Substituted 4-Quinazolinone Derivatives As Hypnotics and Anticonvulsants]". Arzneimittel-Forschung (in German). 13: 688–701. PMID 14085923.