Glutamine
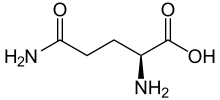 Skeletal formula of L-glutamine | |||
| |||
 | |||
| Names | |||
|---|---|---|---|
| IUPAC name
Glutamine | |||
| Other names
L-Glutamine (levo)glutamide 2,5-Diamino-5-oxopentanoic acid 2-Amino-4-carbamoylbutanoic acid Endari[1] | |||
| Identifiers | |||
CAS Number |
|||
3D model (JSmol) |
| ||
| Abbreviations | Gln, Q | ||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| EC Number |
| ||
| KEGG | |||
PubChem CID |
|||
| UNII | |||
InChI
| |||
SMILES
| |||
| Properties[2] | |||
Chemical formula |
C5H10N2O3 | ||
| Molar mass | 146.146 g·mol−1 | ||
| Melting point | decomposes around 185°C | ||
Solubility in water |
soluble | ||
| Acidity (pKa) | 2.2 (carboxyl), 9.1 (amino) | ||
Chiral rotation ([α]D) |
+6.5º (H2O, c = 2) | ||
| Pharmacology | |||
| A16AA03 (WHO) | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
| Names | |
|---|---|
| Trade names | Endari, Nutrestore |
IUPAC name
| |
| Clinical data | |
| Drug class | Gastrointestinal agent |
| WHO AWaRe | UnlinkedWikibase error: ⧼unlinkedwikibase-error-statements-entity-not-set⧽ |
| Pregnancy category |
|
| Routes of use | By mouth |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a617035 |
| Legal | |
| License data | |
| Legal status |
|
| Identifiers | |
| CAS Number |
|
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| PDB ligand | |
| ATC code | |
| Chemical and physical data | |
| Formula | C5H10N2O3 |
| Molar mass | 146.15 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
Glutamine (symbol Gln or Q)[4] is an α-amino acid that is used in the biosynthesis of proteins. Its side chain is similar to that of glutamic acid, except the carboxylic acid group is replaced by an amide. It is classified as a charge-neutral, polar amino acid. It is non-essential and conditionally essential in humans, meaning the body can usually synthesize sufficient amounts of it, but in some instances of stress, the body's demand for glutamine increases, and glutamine must be obtained from the diet.[5][6] It is encoded by the codons CAA and CAG.
In human blood, glutamine is the most abundant free amino acid.[7]
The dietary sources of glutamine includes especially the protein-rich foods like beef, chicken, fish, dairy products, eggs, vegetables like beans, beets, cabbage, spinach, carrots, parsley, vegetable juices and also in wheat, papaya, Brussels sprouts, celery, kale and fermented foods like miso.
Functions
Glutamine plays a role in a variety of biochemical functions:
- Protein synthesis, as any other of the 20 proteinogenic amino acids
- Lipid synthesis, especially by cancer cells.[8]
- Regulation of acid-base balance in the kidney by producing ammonium[9]
- Cellular energy, as a source, next to glucose[10]
- Nitrogen donation for many anabolic processes, including the synthesis of purines[7]
- Carbon donation, as a source, refilling the citric acid cycle[11]
- Nontoxic transporter of ammonia in the blood circulation.
- Integrity of healthy intestinal mucosa, though small randomized trials have shown no benefit in Crohn’s disease.[12]
Production
Glutamine is produced industrially using mutants of Brevibacterium flavum, which gives ca. 40 g/L in 2 days using glucose as a carbon source.[13] Glutamine is synthesized by the enzyme glutamine synthetase from glutamate and ammonia. The most relevant glutamine-producing tissue is the muscle mass, accounting for about 90% of all glutamine synthesized. Glutamine is also released, in small amounts, by the lungs and brain.[14] Although the liver is capable of relevant glutamine synthesis, its role in glutamine metabolism is more regulatory than producing, since the liver takes up large amounts of glutamine derived from the gut.[7]
Consumers
The most eager consumers of glutamine are the cells of intestines,[7] the kidney cells for the acid-base balance, activated immune cells,[15] and many cancer cells.[8][11][16]
Uses
Nutrition
Glutamine is the most abundant naturally occurring, nonessential amino acid in the human body, and one of the few amino acids that can directly cross the blood–brain barrier.[7] Humans obtain glutamine through catabolism of proteins in foods they eat.[17] In states where tissue is being built or repaired, like growth of babies, or healing from wounds or severe illness, glutamine becomes conditionally essential.[17]
Sickle cell disease
In 2017, the U.S. Food and Drug Administration (FDA) approved L-glutamine oral powder, marketed as Endari, to reduce severe complications of sickle cell disease in people aged five years and older with the disorder.[1]
The safety and efficacy of L-glutamine oral powder were studied in a randomized trial of subjects ages five to 58 years old with sickle cell disease who had two or more painful crises within the 12 months prior to enrollment in the trial.[1] Subjects were assigned randomly to treatment with L-glutamine oral powder or placebo, and the effect of treatment was evaluated over 48 weeks.[1] Subjects who were treated with L-glutamine oral powder experienced fewer hospital visits for pain treated with a parenterally administered narcotic or ketorolac (sickle cell crises), on average, compared to subjects who received a placebo (median 3 vs. median 4), fewer hospitalizations for sickle cell pain (median 2 vs. median 3), and fewer days in the hospital (median 6.5 days vs. median 11 days).[1] Subjects who received L-glutamine oral powder also had fewer occurrences of acute chest syndrome (a life-threatening complication of sickle cell disease) compared with patients who received a placebo (8.6 percent vs. 23.1 percent).[1]
Common side effects of L-glutamine oral powder include constipation, nausea, headache, abdominal pain, cough, pain in the extremities, back pain and chest pain.[1]
L-glutamine oral powder received orphan drug designation.[1] The FDA granted the approval of Endari to Emmaus Medical Inc.[1]
Medical food
Glutamine is marketed as medical food and is prescribed when a medical professional believes a person in their care needs supplementary glutamine due to metabolic demands beyond what can be met by endogenous synthesis or diet.[18]
Safety
Glutamine is safe in adults and in preterm infants.[19] Although glutamine is metabolized to glutamate and ammonia, both of which have neurological effects, their concentrations are not increased much, and no adverse neurological effects were detected.[19] The observed safe level for supplemental L-glutamine in normal healthy adults is 14 g/day.[20]
Adverse effects of glutamine have been prescribed for people receiving home parenteral nutrition and those with liver-function abnormalities.[21] Although glutamine has no effect on the proliferation of tumor cells, it is still possible that glutamine supplementation may be detrimental in some cancer types.[22]
Ceasing glutamine supplementation in people adapted to very high consumption may initiate a withdrawal effect, raising the risk of health problems such as infections or impaired integrity of the intestine.[22]
Structure
Glutamine can exist in either of two enantiomeric forms, L-glutamine and D-glutamine. The L-form is found in nature. Glutamine contains an α-amino group which is in the protonated −NH3+ form under biological conditions and a carboxylic acid group which is in the deprotonated −COO− form, known as carboxylate, under physiological conditions.

Research

Glutamine mouthwash may be useful to prevent oral mucositis in people undergoing chemotherapy but intravenous glutamine does not appear useful to prevent mucositis in the GI tract.[24]
Glutamine supplementation was thought to have potential to reduce complications in people who are critically ill or who have had abdominal surgery but this was based on poor quality clinical trials.[25] Supplementation does not appear to be useful in adults or children with Crohn's disease or inflammatory bowel disease, but clinical studies as of 2016 were underpowered.[12] Supplementation does not appear to have an effect in infants with significant problems of the stomach or intestines.[26]
Some athletes use L-glutamine as supplement. Studies support the positive effects of the chronic oral administration of the supplement on the injury and inflammation induced by intense aerobic and exhaustive exercise, but the effects on muscle recovery from weight training are unclear.[27]
See also
References
- 1 2 3 4 5 6 7 8 9 "FDA approves new treatment for sickle cell disease". U.S. Food and Drug Administration (FDA) (Press release). 7 July 2017. Archived from the original on 14 December 2019. Retrieved 10 July 2017.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ↑ Weast, Robert C., ed. (1981). CRC Handbook of Chemistry and Physics (62nd ed.). Boca Raton, FL: CRC Press. p. C-311. ISBN 0-8493-0462-8..
- ↑ "Glutamine Use During Pregnancy". Drugs.com. 30 September 2019. Archived from the original on 3 August 2020. Retrieved 23 April 2020.
- ↑ "Nomenclature and Symbolism for Amino Acids and Peptides". IUPAC-IUB Joint Commission on Biochemical Nomenclature. 1983. Archived from the original on 9 October 2008. Retrieved 5 March 2018.
- ↑ Dietary Reference Intakes: The Essential Guide to Nutrient Requirements, published by the Institute of Medicine's Food and Nutrition Board, currently available online at "Archived copy". Archived from the original on 5 July 2014. Retrieved 14 July 2014.
{{cite web}}: CS1 maint: archived copy as title (link) - ↑ Lacey, JM; Wilmore, DW (August 1990). "Is glutamine a conditionally essential amino acid?". Nutrition Reviews. 48 (8): 297–309. doi:10.1111/j.1753-4887.1990.tb02967.x. PMID 2080048.
- 1 2 3 4 5 Brosnan, John T. (June 2003). "Interorgan amino acid transport and its regulation". J. Nutr. 133 (6 Suppl 1): 2068S–2072S. doi:10.1093/jn/133.6.2068S. PMID 12771367.

- 1 2 Corbet C, Feron O (2015). "Metabolic and mind shifts: from glucose to glutamine and acetate addictions in cancer". Current Opinion in Clinical Nutrition and Metabolic Care. 18 (4): 346–353. doi:10.1097/MCO.0000000000000178. PMID 26001655. S2CID 1478014.
- ↑ Hall, John E.; Guyton, Arthur C. (2006). Textbook of Medical Physiology (11th ed.). St. Louis, Mo: Elsevier Saunders. p. 393. ISBN 978-0-7216-0240-0.
- ↑ Aledo, J. C. (2004). "Glutamine breakdown in rapidly dividing cells: Waste or investment?". BioEssays. 26 (7): 778–785. doi:10.1002/bies.20063. PMID 15221859.
- 1 2 Yuneva, M.; Zamboni, N.; Oefner, P.; Sachidanandam, R.; Lazebnik, Y. (2007). "Deficiency in glutamine but not glucose induces MYC-dependent apoptosis in human cells". The Journal of Cell Biology. 178 (1): 93–105. doi:10.1083/jcb.200703099. PMC 2064426. PMID 17606868.
- 1 2 Yamamoto, T; Shimoyama, T; Kuriyama, M (8 December 2016). "Dietary and enteral interventions for Crohn's disease". Current Opinion in Biotechnology. 44: 69–73. doi:10.1016/j.copbio.2016.11.011. PMID 27940405.
- ↑ Karlheinz Drauz, Ian Grayson, Axel Kleemann, Hans-Peter Krimmer, Wolfgang Leuchtenberger, Christoph Weckbecker (2007). "Amino Acids". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a02_057.pub2.
{{cite encyclopedia}}: CS1 maint: multiple names: authors list (link) - ↑ Newsholme, P.; Lima, M. M. R.; Procopio, J.; Pithon-Curi, T. C.; Doi, S. Q.; Bazotte, R. B.; Curi, R. (2003). "Glutamine and glutamate as vital metabolites". Brazilian Journal of Medical and Biological Research. 36 (2): 153–163. doi:10.1590/S0100-879X2003000200002. PMID 12563517.
- ↑ Newsholme, P. (2001). "Why is L-glutamine metabolism important to cells of the immune system in health, postinjury, surgery or infection?". The Journal of Nutrition. 131 (9 Suppl): 2515S–2522S, discussion 2522S–4S. doi:10.1093/jn/131.9.2515S. PMID 11533304.
- ↑ Fernandez-de-Cossio-Diaz, Jorge; Vazquez, Alexei (18 October 2017). "Limits of aerobic metabolism in cancer cells". Scientific Reports. 7 (1): 13488. Bibcode:2017NatSR...713488F. doi:10.1038/s41598-017-14071-y. ISSN 2045-2322. PMC 5647437. PMID 29044214.
- 1 2 Watford, Malcolm (September 2015). "Glutamine and glutamate: Nonessential or essential amino acids?". Animal Nutrition. 1 (3): 119–122. doi:10.1016/j.aninu.2015.08.008. PMC 5945979. PMID 29767158.
- ↑ "GlutaSolve, NutreStore, SYMPT-X G.I., SYMPT-X Glutamine (glutamine) Drug Side Effects, Interactions, and Medication Information on eMedicineHealth". eMedicineHealth. Archived from the original on 2 February 2017. Retrieved 24 January 2017.
- 1 2 Garlick PJ (2001). "Assessment of the safety of glutamine and other amino acids". The Journal of Nutrition. 131 (9 Suppl): 2556S–61S. doi:10.1093/jn/131.9.2556S. PMID 11533313.
- ↑ Shao A, Hathcock JN (2008). "Risk assessment for the amino acids taurine, L-glutamine and L-arginine". Regulatory Toxicology and Pharmacology. 50 (3): 376–99. doi:10.1016/j.yrtph.2008.01.004. PMID 18325648.
- ↑ Buchman AL (2001). "Glutamine: commercially essential or conditionally essential? A critical appraisal of the human data". The American Journal of Clinical Nutrition. 74 (1): 25–32. doi:10.1093/ajcn/74.1.25. PMID 11451714.
- 1 2 Holecek M (2013). "Side effects of long-term glutamine supplementation". Journal of Parenteral and Enteral Nutrition. 37 (5): 607–16. doi:10.1177/0148607112460682. PMID 22990615.
- ↑ Stehle P, Kuhn KS (2015). "Glutamine: an obligatory parenteral nutrition substrate in critical care therapy". Biomed Res Int. 2015: 1–7. doi:10.1155/2015/545467. PMC 4606408. PMID 26495301.
- ↑ Berretta, M; Michieli, M; Di Francia, R; Cappellani, A; Rupolo, M; Galvano, F; Fisichella, R; Berretta, S; Tirelli, U (1 January 2013). "Nutrition in oncologic patients during antiblastic treatment". Frontiers in Bioscience. 18: 120–32. doi:10.2741/4091. PMID 23276913. Archived from the original on 3 August 2020. Retrieved 23 September 2021.
- ↑ Tao, KM; Li, XQ; Yang, LQ; Yu, WF; Lu, ZJ; Sun, YM; Wu, FX (9 September 2014). "Glutamine supplementation for critically ill adults". The Cochrane Database of Systematic Reviews (9): CD010050. doi:10.1002/14651858.CD010050.pub2. PMC 6517119. PMID 25199493.
- ↑ Moe-Byrne, T; Brown, JV; McGuire, W (18 April 2016). McGuire, William (ed.). "Glutamine supplementation to prevent morbidity and mortality in preterm infants" (PDF). The Cochrane Database of Systematic Reviews. 4: CD001457. doi:10.1002/14651858.CD001457.pub6. PMC 7055588. PMID 27089158. Archived (PDF) from the original on 26 April 2021. Retrieved 23 September 2021.
- ↑ Raizel, Raquel; Tirapegui, Julio (5 December 2018). "Role of glutamine, as free or dipeptide form, on muscle recovery from resistance training: a review study". Nutrire. 43 (1): 28. doi:10.1186/s41110-018-0087-9. ISSN 2316-7874. S2CID 81105808.
External links
- Glutamine spectra Archived 31 January 2018 at the Wayback Machine acquired through mass spectroscopy
- "Glutamine". Drug Information Portal. U.S. National Library of Medicine. Archived from the original on 2 October 2021. Retrieved 23 September 2021.