TPA-023
TPA-023 (MK-0777) is an anxiolytic drug with a novel chemical structure, which is used in scientific research. It has similar effects to benzodiazepine drugs, but is structurally distinct and so is classed as a nonbenzodiazepine anxiolytic. It is a subtype-selective, mixed allosteric modular at the benzodiazepine location on GABAA receptors, where it acts as a partial agonist at the α2 and α3 subtypes, but as a silent antagonist at α1 and α5 subtypes.[1] It has primarily anxiolytic and anticonvulsant effects in animal tests, but with no sedative effects even at 50 times the effective anxiolytic dose.[2][3]
 | |
 | |
| Clinical data | |
|---|---|
| Other names | MK-0777 |
| Routes of administration | By mouth |
| Pharmacokinetic data | |
| Metabolism | liver |
| Elimination half-life | 6.7 hours |
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
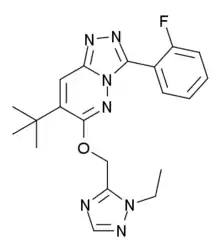
| Formula | C20H22FN7O |
| Molar mass | 395.442 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
| (verify) | |
In human trials on healthy volunteers, TPA-023 was comparable to lorazepam, but had much less side effects on cognition, memory, alertness or coordination.[4] In Phase II trials, the compound was significantly superior to placebo without inducing sedation. The clinical development was halted due to preclinical toxicity (cataract) in long term dosing studies.[5][6] TPA-023 is well absorbed following oral administration and extensively metabolised by the liver, with a half-life of 6.7 hours.[7] The main enzyme involved in its metabolism is CYP3A4, with some contribution by CYP3A5.[8]
References
- Kohut SJ, Ator NA (July 2008). "Novel discriminative stimulus effects of TPA023B, subtype-selective gamma-aminobutyric-acid(A)/benzodiazepine modulator: comparisons with zolpidem, lorazepam, and TPA023". Pharmacology, Biochemistry, and Behavior. 90 (1): 65–73. doi:10.1016/j.pbb.2008.02.019. PMC 3010402. PMID 18395780.
- Carling RW, Madin A, Guiblin A, Russell MG, Moore KW, Mitchinson A, et al. (November 2005). "7-(1,1-Dimethylethyl)-6-(2-ethyl-2H-1,2,4-triazol-3-ylmethoxy)-3-(2-fluorophenyl)-1,2,4-triazolo[4,3-b]pyridazine: a functionally selective gamma-aminobutyric acid(A) (GABA(A)) alpha2/alpha3-subtype selective agonist that exhibits potent anxiolytic activity but is not sedating in animal models". Journal of Medicinal Chemistry. 48 (23): 7089–92. doi:10.1021/jm058034a. PMID 16279764.
- Atack JR, Wafford KA, Tye SJ, Cook SM, Sohal B, Pike A, et al. (January 2006). "TPA023 [7-(1,1-dimethylethyl)-6-(2-ethyl-2H-1,2,4-triazol-3-ylmethoxy)-3-(2-fluorophenyl)-1,2,4-triazolo[4,3-b]pyridazine], an agonist selective for alpha2- and alpha3-containing GABAA receptors, is a nonsedating anxiolytic in rodents and primates". The Journal of Pharmacology and Experimental Therapeutics. 316 (1): 410–22. doi:10.1124/jpet.105.089920. PMID 16183706. S2CID 23047072.
- de Haas SL, de Visser SJ, van der Post JP, de Smet M, Schoemaker RC, Rijnbeek B, et al. (June 2007). "Pharmacodynamic and pharmacokinetic effects of TPA023, a GABA(A) alpha(2,3) subtype-selective agonist, compared to lorazepam and placebo in healthy volunteers". Journal of Psychopharmacology. 21 (4): 374–83. doi:10.1177/0269881106072343. PMID 17092968. S2CID 22626040.
- Möhler H (June 2011). "The rise of a new GABA pharmacology". Neuropharmacology. 60 (7–8): 1042–9. doi:10.1016/j.neuropharm.2010.10.020. PMID 21035473. S2CID 46645932.
- Atack JR (2008). "GABA(A) receptor subtype-selective efficacy: TPA023, an alpha2/alpha3 selective non-sedating anxiolytic and alpha5IA, an alpha5 selective cognition enhancer". CNS Neuroscience & Therapeutics. 14 (1): 25–35. doi:10.1111/j.1527-3458.2007.00034.x. PMC 6494020. PMID 18482097.
- Polsky-Fisher SL, Vickers S, Cui D, Subramanian R, Arison BH, Agrawal NG, et al. (June 2006). "Metabolism and disposition of a potent and selective GABA-Aalpha2/3 receptor agonist in healthy male volunteers". Drug Metabolism and Disposition. 34 (6): 1004–11. doi:10.1124/dmd.105.008193. PMID 16510541. S2CID 17373.
- Ma B, Polsky-Fisher SL, Vickers S, Cui D, Rodrigues AD (August 2007). "Cytochrome P450 3A-dependent metabolism of a potent and selective gamma-aminobutyric acid Aalpha2/3 receptor agonist in vitro: involvement of cytochrome P450 3A5 displaying biphasic kinetics". Drug Metabolism and Disposition. 35 (8): 1301–7. doi:10.1124/dmd.107.014753. PMID 17460031. S2CID 86847445.