Propan-1-ol
Propan-1-ol (also propanol, n-propyl alcohol) is a primary alcohol with the formula CH3CH2CH2OH and sometimes represented as PrOH or n-PrOH. It is a colorless liquid and an isomer of 2-propanol. It is formed naturally in small amounts during many fermentation processes and used as a solvent in the pharmaceutical industry, mainly for resins and cellulose esters, and, sometimes, as a disinfecting agent.
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Propan-1-ol[1] | |
Other names
| |
| Identifiers | |
CAS Number |
|
3D model (JSmol) |
|
| 3DMet | |
Beilstein Reference |
1098242 |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.000.679 |
| EC Number |
|
Gmelin Reference |
25616 |
| KEGG | |
| MeSH | 1-Propanol |
PubChem CID |
|
| RTECS number |
|
| UNII | |
| UN number | 1274 |
CompTox Dashboard (EPA) |
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula |
C3H8O |
| Molar mass | 60.096 g·mol−1 |
| Appearance | Colorless liquid |
| Odor | mild, alcohol-like[2] |
| Density | 0.803 g/mL |
| Melting point | −126 °C; −195 °F; 147 K |
| Boiling point | 97 to 98 °C; 206 to 208 °F; 370 to 371 K |
Solubility in water |
miscible |
| log P | 0.329 |
| Vapor pressure | 1.99 kPa (at 20 °C) |
| Acidity (pKa) | 16 |
| Basicity (pKb) | −2 |
Magnetic susceptibility (χ) |
−45.176·10−6 cm3/mol |
Refractive index (nD) |
1.387 |
| Viscosity | 1.959 mPa·s (at 25 °C) [3] |
Dipole moment |
1.68 D |
| Thermochemistry | |
Heat capacity (C) |
143.96 J/(K·mol) |
Std molar entropy (S⦵298) |
192.8 J/(K·mol) |
Std enthalpy of formation (ΔfH⦵298) |
−302.79…−302.29 kJ/mol |
Std enthalpy of combustion (ΔcH⦵298) |
−2.02156…−2.02106 MJ/mol |
| Pharmacology | |
| D08AX03 (WHO) | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards |
Flammable liquid |
| GHS labelling: | |
Pictograms |
  |
Signal word |
Danger |
Hazard statements |
H225, H302, H318, H336 |
Precautionary statements |
P210, P261, P280, P305+P351+P338 |
| NFPA 704 (fire diamond) | |
| Flash point | 22 °C (72 °F; 295 K) |
Autoignition temperature |
371 °C (700 °F; 644 K) |
| Explosive limits | 2.2–13.7%[2] |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose) |
2800 mg/kg (rabbit, oral) 1699 mg/kg (mouse, oral)[4] 1870 mg/kg (rat, oral)[5] |
| NIOSH (US health exposure limits): | |
PEL (Permissible) |
TWA 200 ppm (500 mg/m3)[2] |
REL (Recommended) |
TWA 200 ppm (500 mg/m3) ST 250 ppm (625 mg/m3) [skin][2] |
IDLH (Immediate danger) |
800 ppm[2] |
| Related compounds | |
Related compounds |
Propane Isopropyl alcohol Propanamine Ethanol Butanol |
| Supplementary data page | |
| 1-Propanol (data page) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Chemical properties
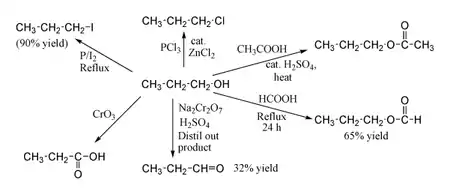
Propan-1-ol shows the normal reactions of a primary alcohol. Thus it can be converted to alkyl halides; for example red phosphorus and iodine produce n-propyl iodide in 80% yield, while PCl3 with catalytic ZnCl2 gives n-propyl chloride. Reaction with acetic acid in the presence of an H2SO4 catalyst under Fischer esterification conditions gives propyl acetate, while refluxing propanol overnight with formic acid alone can produce propyl formate in 65% yield. Oxidation of propan-1-ol with Na2Cr2O7 and H2SO4 gives a 36% yield of propionaldehyde, and therefore for this type of reaction higher yielding methods using PCC or the Swern oxidation are recommended. Oxidation with chromic acid yields propionic acid.
Preparation
Propan-1-ol is manufactured by catalytic hydrogenation of propionaldehyde. Propionaldehyde is produced via the oxo process by hydroformylation of ethylene using carbon monoxide and hydrogen in the presence of a catalyst such as cobalt octacarbonyl or a rhodium complex.[6]
- H2C=CH2 + CO + H2 → CH3CH2CH=O
- CH3CH2CH=O + H2 → CH3CH2CH2OH
A traditional laboratory preparation of propan-1-ol involves treating n-propyl iodide with moist Ag2O.
Safety
Propan-1-ol is thought to be similar to ethanol in its effects on the human body, but 2–4 times more potent. Oral LD50 in rats is 1870 mg/kg (compared to 7060 mg/kg for ethanol). It is metabolized into propionic acid. Effects include alcoholic intoxication and high anion gap metabolic acidosis. As of 2011, one case of lethal propan-1-ol poisoning was reported.[7]
Propan-1-ol as fuel
Propan-1-ol has high octane number and is suitable for engine fuel usage. However, propanol is too expensive to use as a motor fuel. The research octane number (RON) of propanol is 118, and anti-knock index (AKI) is 108.[8]
References
- Favre HA, Powell WH (2014). Nomenclature of Organic Chemistry: IUPAC Recommendations and Preferred Names 2013 (Blue Book). Cambridge: The Royal Society of Chemistry. p. 61. doi:10.1039/9781849733069. ISBN 9780854041824.
- NIOSH Pocket Guide to Chemical Hazards. "#0533". National Institute for Occupational Safety and Health (NIOSH).
- Pal A, Gaba R (2008). "Volumetric, acoustic, and viscometric studies of molecular interactions in binary mixtures of dipropylene glycol dimethyl ether with 1-alkanols at 298.15 K". J. Chem. Thermodyn. 40 (5): 818–828. doi:10.1016/j.jct.2008.01.008.
- Propan-1-ol toxicity
- "n-Propyl alcohol". Immediately Dangerous to Life or Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- Papa AJ (2011). "Propanols". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a22_173.pub2. ISBN 9783527303854.
- Unmack JL (2011). "N-PROPANOL Health-Base Assessment and Recommendation for HEAC" (PDF).
- "Bioalcohols". Biofuel.org.uk. 2010. Retrieved 16 Apr 2014.
Further reading
- Furniss, B. S.; Hannaford, A. J.; Smith, P. W. G.; Tatchell, A. R. (1989), Vogel's Textbook of Practical Organic Chemistry (5th ed.), Harlow: Longman, ISBN 0-582-46236-3
- Lide DR, ed. (2006). CRC Handbook of Chemistry and Physics (87th ed.). TF-CRC. ISBN 0849304873.
- O'Neil MJ, ed. (2006). The Merck Index: An Encyclopedia of Chemicals, Drugs, and Biologicals (14th ed.). Merck. ISBN 091191000X.
- Perkin WH, Kipping FS (1922). Organic Chemistry. London: W. & R. Chambers. ISBN 0080223540.
