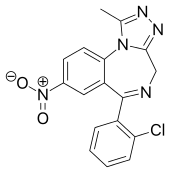
Clonazolam
Clonazolam (also known as clonitrazolam) is a drug of the triazolobenzodiazepine (TBZD) class, which are benzodiazepines (BZDs) fused with a triazole ring. It has had very little research done about its effects and metabolism, and has been sold online as a designer drug.[1][2][3][4][5]
 | |
 | |
| Clinical data | |
|---|---|
| Routes of administration | Oral |
| Legal status | |
| Legal status |
|
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C17H12ClN5O2 |
| Molar mass | 353.77 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
The synthesis of clonazolam was first reported in 1971 and the drug was described as the most active compound in the series tested.[6][7]
Clonazolam is reported to be highly potent and concerns have been raised that it and flubromazolam in particular may pose comparatively higher risk than other designer benzodiazepines due to their ability to produce strong sedation and amnesia at as little as 0.5 mg.[8]
Legality
United Kingdom
In the UK, clonazolam has been classified as a Class C drug by the May 2017 amendment to The Misuse of Drugs Act 1971 along with several other designer benzodiazepine drugs.[9]
United States
It is a Schedule I medicine in Virginia, Louisiana, North Carolina and Oregon and is not FDA approved for human consumption. Virginia State Law has declared all of the following medications are now schedule I: clonazolam, etizolam, flualprazolam, flubromazolam, and flubromazepam.[10] Minnesota declared clonazolam a Schedule I drug in August 2020.
Australia
In Australia, clonazolam is classified as a controlled substance in some states.[11]
Sweden
Sweden's public health agency suggested classifying clonazolam as a hazardous substance on June 1, 2015.[12]
Effects
Clonazolam's effects are similar to other benzodiazepines, such as anxiolysis, disinhibition, lethargy, muscle relaxation, and euphoria. While no dose of clonazolam is considered "safe" due to its lack of research and extreme potency, doses higher than 0.5 mg can cause benzodiazepine overdose in some individuals. The effects of a benzodiazepine overdose include sedation, confusion, insufficient breathing, loss of consciousness, and death. Because dependence can occur in a short period of time, or even with a large initial dose, withdrawal symptoms (including seizures and death) may occur acutely following the period of intoxication.
See also
- Adinazolam
- Alprazolam
- Clonazepam, no triazole ring
- Estazolam, (licensed)
- Flubromazolam
- Pyrazolam
- Triazolam
References
- Huppertz LM, Bisel P, Westphal F, Franz F, Auwärter V, Moosmann B (July 2015). "Characterization of the four designer benzodiazepines clonazolam, deschloroetizolam, flubromazolam, and meclonazepam, and identification of their in vitro metabolites". Forensic Toxicology. 33 (2): 388–395. doi:10.1007/s11419-015-0277-6. S2CID 33278305.
- Meyer MR, Bergstrand MP, Helander A, Beck O (May 2016). "Identification of main human urinary metabolites of the designer nitrobenzodiazepines clonazolam, meclonazepam, and nifoxipam by nano-liquid chromatography-high-resolution mass spectrometry for drug testing purposes". Analytical and Bioanalytical Chemistry. 408 (13): 3571–91. doi:10.1007/s00216-016-9439-6. PMID 27071765. S2CID 25831532.
- Chaslot M, El Balkhi S, Robin T, Morichon J, Picard N, Saint-Marcoux F (June 2016). "Exploration des métabolites de 8 benzodiazépines de synthèse". Toxicologie Analytique et Clinique. 28 (2): S32. doi:10.1016/j.toxac.2016.03.053.
- Pettersson Bergstrand M, Helander A, Hansson T, Beck O (April 2017). "Detectability of designer benzodiazepines in CEDIA, EMIT II Plus, HEIA, and KIMS II immunochemical screening assays". Drug Testing and Analysis. 9 (4): 640–645. doi:10.1002/dta.2003. PMID 27366870.
- Høiseth G, Tuv SS, Karinen R (November 2016). "Blood concentrations of new designer benzodiazepines in forensic cases". Forensic Science International. 268: 35–38. doi:10.1016/j.forsciint.2016.09.006. PMID 27685473.
- Hester JB, Rudzik AD, Kamdar BV (November 1971). "6-phenyl-4H-s-triazolo[4,3-a][1,4]benzodiazepines which have central nervous system depressant activity". Journal of Medicinal Chemistry. 14 (11): 1078–81. doi:10.1021/jm00293a015. PMID 5165540.
- EP 0072029, Borer R, Gerecke M, Kyburz E, "Triazolobenzazepines, process and intermediates for their preparation and medicines containing them", published 22 October 1986, assigned to F Hoffmann La Roche AG
- Moosmann B, King LA, Auwärter V (June 2015). "Designer benzodiazepines: A new challenge". World Psychiatry. 14 (2): 248. doi:10.1002/wps.20236. PMC 4471986. PMID 26043347.
- "The Misuse of Drugs Act 1971 (Amendment) Order 2017".
- "§ 54.1-3446. Schedule I".
- "Controlled substances (controlled drugs, precursors and plants) regulations 2014 - Schedule 1". South Australian Current Regulations. Retrieved 2021-02-12.
- "23 nya ämnen kan klassas som narkotika eller hälsofarlig vara" (in Swedish). Folkhälsomyndigheten. Retrieved 2015-08-06.