CRL-40,940
 | |
| Identifiers | |
|---|---|
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
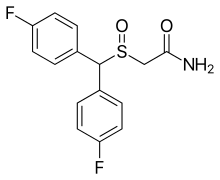
| Formula | C15H13F2NO2S |
| Molar mass | 309.33 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
CRL-40,940 (also known as flmodafinil, bisfluoromodafinil and lauflumide) is the bisfluoro analog of modafinil.[1][2][3][4] It is a eugeroic as well as a weak dopamine reuptake inhibitor. Its inventors claim that it is more effective than modafinil and adrafinil, with fewer side effects. It was patented in 2013, and pre-clinical trials have been underway since December 2015.[5]
See also
- Adrafinil
- Armodafinil
- CE-123
- CRL-40,941
- Fluorenol
References
- ↑ CA 1199916, Lafon L, "Benzhydrylsulfinylacetamide derivatives", published 28 January 1986
- ↑ Cao J, Prisinzano TE, Okunola OM, Kopajtic T, Shook M, Katz JL, Newman AH (October 2010). "Structure-Activity Relationships at the Monoamine Transporters for a Novel Series of Modafinil (2-[(diphenylmethyl)sulfinyl]acetamide) Analogues". ACS Medicinal Chemistry Letters. 2 (1): 48–52. doi:10.1021/ml1002025. PMC 3041981. PMID 21344069.
- ↑ US 20130295196, Konofal E, "Lauflumide and the enantiomers thereof, method for preparing same and therapeutic uses thereof", published 7 November 2013
- ↑ Louis Lafon (18 December 1984). "Patent US 4489095 A - Halogenobenzhydrylsulfinylacetohydroxamic acids". Retrieved 25 July 2015.
- ↑ "Pipeline". NLS Pharmaceuticals.
This article is issued from Offline. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.